Abstract
Background: Functional neuroimaging studies on schizophrenia have suggested abnormal task-related functional connectivity in patients with schizophrenia who have auditory verbal hallucinations (AVHs). However, little is known about intrinsic functional connectivity in these patients.
Methods: Between January 2009 and February 2010, we studied patients with schizophrenia who had persistent and treatment-refractory AVHs in comparison with healthy controls. Using functional magnetic resonance imaging, we studied the functional connectivity of multiple resting state networks (RSNs) and their relation to symptom severity. We analyzed the data using a spatial group independent component analysis, and we used random-effects t tests to compare spatial components between groups.
Results: There were 10 patients and 14 controls enrolled in this study. In total, 16 RSNs were identified, from which we selected 4 networks of interest for further analyses. Within a speech-related network, patients showed increased connectivity in bilateral temporal regions and decreased connectivity in the cingulate cortex. Within 2 additional RSNs associated with attention and executive control, respectively, patients exhibited abnormal connectivity in the precuneus and right lateral prefrontal areas. We found correlations between measures of AVH severity and functional connectivity of the left anterior cingulate, left superior temporal gyrus and right lateral prefrontal cortex.
Limitations: The relatively small sample size, the patients’ use of antipsychotic medication and the lack of a clinical control group have to be considered as potential limitations.
Conclusion: Our findings indicate that disrupted intrinsic connectivity of a speech-related network could underlie persistent AVHs in patients with schizophrenia. In addition, the occurrence of hallucinatory symptoms seems to modulate RSNs associated with attention and executive control.
Introduction
Auditory verbal hallucinations (AVHs), defined as sensory experiences in the absence of a corresponding external stimulus,1 are a core symptom of schizophrenia and related spectrum disorders. Numerous neuroimaging studies have indicated that AVHs are associated with the abnormal structure and function of a widely distributed set of brain regions, such as the lateral prefrontal cortices, the cingulate cortex and regions of the temporal lobes, that are involved in language, attention, executive function and memory.2,3 The most consistent findings have implicated frontotemporal pathways as the main neural substrate underlying AVHs,2,4 leading to cognitive models of AVHs as a result of a functional breakdown in appropriate monitoring of inner speech generation.5,6 On a neural network level, abnormal “corollary discharge,” a mechanism that modulates self-generated responses to distinguish between self-generated and externally generated information, has been proposed as a putative pathophysiologic mechanism of hallucinatory phenomena.7 For instance, dysfunctional efference copy/corollary discharge interactions within speech-related neural networks8 could result in a misattribution of self-generated actions and perceptions, eventually giving rise to AVHs.9,10 Aberrant frontotemporal coupling in patients with schizophrenia has been described in several studies so far,11–13 and, more recently, studies investigating cortico–cortical connectivity in patients who have AVHs have revealed abnormal interactions within speech-related neural pathways.13,14 However, patterns of functional disconnection in patients with schizophrenia may be widespread,15–17 eventually affecting multiple systems with discrete spatial distributions and functions,15–17 including networks subserving attentional control, memory, executive function and self-referential processing. At present, however, it is unclear if and how these networks contribute to the pathophysiology of AVHs.
In this study, we investigated the functional neuroanatomy of AVHs in patients with schizophrenia using a resting-state functional connectivity approach.18 Given the possibility that task-driven protocols may reveal activation patterns driven by interactions between experimental stimuli and AVHs,4,14,19,20 we chose to investigate symptom-related patterns of dysconnectivity in the absence of experimentally controlled stimulation. In this regard, resting-state functional connectivity methods essentially aim to identify neural networks characterized by ongoing spontaneous modulations of the blood oxygen level–dependent (BOLD) signal in the absence of specific task-related activity.21,22 One of the most extensively investigated resting-state networks (RSNs) so far is a set of brain regions that consistently exhibits activity decreases during cognitively demanding tasks. This RSN has been referred to as the “default mode network” (DMN),22,23 a functionally heterogeneous network that has been associated with self-referential processing, affective control and episodic memory.23,24 Abnormal DMN activity in patients with schizophrenia has been reported in a number of studies so far.16,25,26 Apart from the DMN, however, several other RSNs, including lateral frontoparietal and medial-frontal systems, have now been consistently identified.22,27,28 These RSNs have been suggested to closely correspond to a wide range of cognitive processes, such as language, attention, memory and executive control.28–30
We used independent component analysis, a multivariate statistical approach to resting-state data, to assess the functional connectivity of multiple RSNs in patients with chronic and treatment-resistant AVHs compared with healthy controls. Independent component analysis is a technique that maximizes the independence between output components,31 thus identifying a set of spatially nonoverlapping and temporally coherent networks by measuring functional covariance patterns between different brain areas.32 Specifically, we investigated 4 anatomically distinct RSNs of interest that have been previously associated with the pathophysiology of AVHs: RSNs related to speech processing, attention, executive control and DMN function.2,4,33 Within these networks, we predicted symptom-related functional connectivity of the temporal cortex and disrupted connectivity of regions associated with cognitive control, such as the anterior cingulate and the lateral prefrontal cortices. In addition, we explored the relation between functional connectivity and symptom severity, as assessed by AVH-specific psychometric scores.
Methods
Participants
We recruited patients with schizophrenia from among the in-and outpatients treated at the Departments of Psychiatry II (Günzburg) and III (Ulm) at the University of Ulm, Germany. In addition to a detailed interview conducted by experienced clinical psychiatrists (R.C.W., N.D.W.), case notes were reviewed to corroborate a definitive diagnosis. All patients met DSM-IV criteria for schizophrenia, paranoid subtype. We excluded patients from participation if they had a current axis-I mood, substance-related or anxiety disorder or a concurrent axis-II disorder according to DSM-IV; had a history of dependence on illicit drugs and alcohol; had an insufficient command of the German language; and had sensorimotor deficits or other neurologic disorders.
Patients included in this study had to meet several specific inclusion criteria. First, they must have had medication-resistant AVHs, defined by persistent hallucinations in the presence of at least 2 previous clinically ineffective drug trials (each treatment period > 6 wk) with different antipsychotics at an adequate dosage. Second, they must not have shown pronounced formal thought disorder symptoms. Third, they must have had sufficient insight into their hallucinatory experiences and ability to provide detailed self-reports about their AVHs before functional magnetic resonance imaging (fMRI) and during a postscanning exit interview.
Patients’ ability to provide informed consent was determined after an interview conducted by 2 experienced clinical psychiatrists (R.C.W., N.D.W.), where the background, the aims and the experimental procedures of the study were presented and discussed in detail. After the interview, patients were asked to briefly summarize the study’s scope and procedures and were given sufficient time to address study-specific questions.
We rated general psychopathology using the Brief Psychiatric Rating Scale (BPRS)34 and the Positive and Negative Syndrome Scale (PANSS).35 We assessed the severity of AVHs using the auditory hallucinations scale (AHS) sum score, as provided by the Psychotic Symptoms Rating Scales (PsyRatS).37
The healthy control group was recruited from the University of Ulm campus and from the University Department of Psychiatry III. Controls were matched for age, education and handedness. We excluded controls from participation if they had a current axis-I or a concurrent axis-II disorder according to DSM-IV, had a first-degree relative with a neurologic or psychiatric disorder, had a history of dependence on illicit drugs and alcohol, were currently taking any psychotropic medication, and had sensorimotor deficits or other neurologic disorders.
The study was approved by the local research ethics committee (University of Ulm, Germany). All experimental procedures were carried out with the informed and written consent of the participants.
Imaging data acquisition
The functional data were acquired using a 3-T Magnetom ALLEGRA head MRI system (Siemens) at the Department of Psychiatry and Psychotherapy III at the University of Ulm, Germany. Scans were performed in darkness, and the participants were explicitly instructed to relax without falling asleep, keep their eyes closed, not think about anything in particular and move as little as possible. Adherence to these instructions was verified by verbal contact immediately after the resting-state scan and as part of a postscanning exit interview. In this interview, patients reported the occurrence of AVHs during the MRI session (self-reported AVH occurring several times during the resting-state scan over time periods of several seconds).
We obtained T2*-weighted images using echo-planar imaging in an axial orientation (repetition time 2000 ms, echo time 30 ms, field of view 192 mm, flip angle 80°, voxel size 3 × 3 × 3 mm, 33 slices, slice thickness 3 mm, gap 1 mm). Within a session, 180 whole-brain volumes were acquired. Prior to data processing, the first 8 volumes of the time series were discarded to account for MRI equilibration effects.
Data analysis
Data preprocessing was performed with SPM5 (Wellcome Trust Centre for Neuroimaging) and MATLAB 7.3 (Math-Works). The functional images were corrected for motion artifacts and spatially normalized to the SPM5 EPI standard template. All images were spatially smoothed with a 9-mm full-width at half-maximum isotropic Gaussian kernel. We performed a spatial independent component analysis including both patients and controls using the “Group ICA of fMRI Toolbox” (GIFT; http://mialab.mrn.org/software/gift/index.html).38 The dimensionality of the functional data for each participant was reduced using 3 consecutive steps of principal component analysis alternated with data linked across participants, resulting in one aggregate mixing matrix for all participants. We performed an independent component analysis decomposition using the Infomax algorithm to extract 16 independent components consisting of group spatial maps and related time courses. We used “minimum description length” criteria to estimate the order selection (i.e., the number of independent components from the smoothed data sets after taking into account the spatial and temporal correlation of the fMRI data).39 The estimated independent components were used for a back reconstruction into individual components using the aggregate mixing matrix created during the dimensionality data reduction steps. Data normalization was performed using a Z-score transformation. The individual components consisting of spatial independent maps and time courses were eventually sorted using a priori masks comprising mediolateral prefrontal and cingulate regions, as defined by the Automatic Anatomic Labelling (AAL) Atlas.40 We used 2 masks for spatial sorting: first, as described by other groups,26 we computed a DMN mask comprising the posterior parietal cortex (Brodmann area [BA] 7), frontopolar cortex (BA 10), posterior cingulate cortex, precuneus and occipitoparietal junction (BA 39). Second, to identify networks previously associated with language, attention and cognitive control,16,28,32 we computed a “prefrontal” mask comprising the superior, middle and inferior frontal cortices and the anterior cingulate cortex. We chose 1 component of interest (COI) that showed the highest spatial correlation with the DMN mask and 3 COIs that showed the highest spatial correlation with the prefrontal mask for the second-level within- and between-group analyses (see also the section on Functional connectivity within-group analyses in Results). In addition, we identified an a priori “control” COI that has been previously identified to show a high spatial overlap with primary and secondary visual areas.28 This visual component was identified by inspection of the data using the components output obtained from the independent component analysis and a subsequent within-group second-level analysis (Appendix 1, available at www.cma.ca/jpn). For the visual network, we did not expect symptom- or disease-related connectivity differences in contrast to the other networks of interest.
For each participant’s spatial COI, we used the voxel weights as random-effects variables and analyzed them using SPM5. For within-group analyses, we used voxel-wise 1-sample t tests against the null hypothesis of zero magnitude to calculate within-group maps for each COI. The statistical thresholds for these analyses were set at p < 0.001, uncorrected at the voxel level, and p < 0.05, corrected for spatial extent. On the second level, we compared spatial maps between controls and patients using 2-sample t tests. To fully include those COIs revealed in both groups, we masked these between-group comparisons with a combined mask, which was created as follows. First, we computed 1-sample t tests per RSN and group. Second, thresholded t maps (p < 0.005) were binarized using the “AND” Boolean operator, thus producing binary masks of the combined effect of each diagnostic group. We eventually used these combined spatial maps to explicitly mask the between-group comparisons computed for each RSN. Thresholds of p < 0.001, uncorrected at the voxel level, and p < 0.05, corrected for spatial extent, were chosen for all second between-group comparisons. All anatomic regions and denominations are reported according to the atlases of Talairach and Tournoux.41 Coordinates are maxima in a given cluster according to the Montreal Neurological Institute (MNI) template.
Correlations between functional connectivity indices and psychometric measures
We calculated correlation analyses between indices of functional connectivity and psychometric measures. We computed Spearman correlations using the β parameters (“connectivity strength,” corresponding to the mean voxel weights of the COIs) of significant clusters emerging from the between-group comparisons in the independent component analysis, as well as from psychometric variables (PANSS and BPRS scores, PsyRatS total score). We extracted β parameters from clusters of interest using MarsBar 0.41.42 The data were subsequently processed off-line using the Statistica software package (version 6.0, StatSoft Inc.). A nominal significance level of p < 0.05 was defined and adjusted for multiple comparisons by means of a rough false discovery rate (FDR) correction43 according to the 9 different clusters of interest for which correlations were computed (p level adjusted to p < 0.028).
Results
Participants
We enrolled 10 right-handed patients with schizophrenia who had persistent and treatment-refractory AVHs and 14 unmedicated, matched controls. The demographic and clinical characteristics of participants are summarized in Table 1. All patients were on stable doses of antipsychotic medication for at least 2 months before participating in the study. At the time of scanning, antipsychotic treatment included clozapine (175–650 mg/d) as a primary antipsychotic agent, partly in combination with amisulpride (400 mg/d), aripiprazole (15 mg/d), haloperidol (15 mg/d), paliperidone (12 mg/d), quetiapine (800 mg/d) and risperidone (4 mg/d). The mean chlorpromazine equivalence dosage was 457.3 mg (standard deviation [SD] 342.1 mg).
Demographic and clinical characteristics of healthy controls and patients with schizophrenia who have persistent auditory verbal hallucinations
Functional connectivity within-group analyses
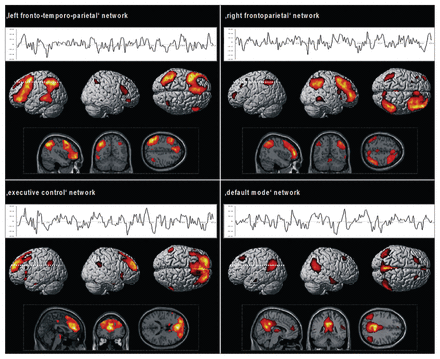
In total, 16 independent components were estimated, consisting of individual spatial independent maps and time courses (Fig. 1). In controls and patients, we identified 4 COIs that were spatially correlated with the a priori prefrontal masks derived from the AAL atlas.40 Consistent with previous research,27,28 we identified the following network patterns. The first COI (left fronto-temporo-parietal,16,28 r = 0.32; Fig. 1, top left) revealed a spatial pattern comprising predominantly the bilateral prefrontal cortex (left lateralized dorso- and ventrolateral prefrontal cortex, medial and superior prefrontal cortex) and left temporoparietal regions. The second COI (right frontoparietal,16,28 r = 0.33; Fig. 1, top right) revealed a network comprising right lateralized ventro- and dorsolateral prefrontal regions, superior and inferior parietal areas, the dorsal and posterior cingulate cortex and the precuneus. The third COI (executive control,28,32 r = 0.22; Fig. 1, bottom left) comprised the bilateral ventrolateral prefrontal cortex, anterior prefrontal regions, cingulate cortex and bilateral middle temporal and inferior parietal areas. The fourth COI (DMN, r = 0.59; Fig. 1, bottom right) showed a pattern of predominantly cortical midline regions, as described in detail by previous studies of DMN function.28,44 The visual network included the primary and secondary visual areas (Appendix 1).
Spatial pattern and related time courses of the resting-state network of interest identified by the group independent component analysis. Results from the second-level within-group analyses including controls and patients with schizophrenia (p < 0.001, uncorrected at the voxel level, p < 0.05 corrected for spatial extent).
Functional connectivity between-group analyses
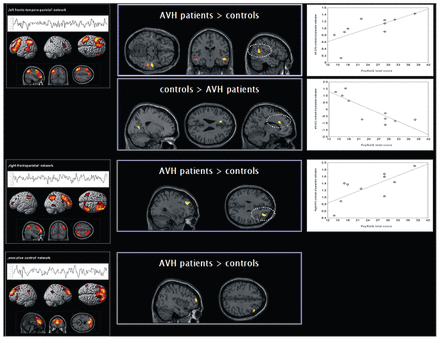
The second-level 2-sample t tests revealed the following findings. Within the left fronto-temporo-parietal RSN, the patient group showed less connectivity in the left anterior cingulate cortex (ACC) and the right posterior cingulate cortex. We found increased connectivity in the left middle and superior temporal gyrus (MTG/STG) and in the right MTG in patients compared with controls (Fig. 2, Table 2). Within the right frontoparietal RSN, the patient group showed increased connectivity in the right middle frontal gyrus (MFG; Fig. 2, Table 2). Within the executive control RSN, patients showed less connectivity in the left precuneus and increased connectivity in the right MFG and superior frontal gyrus (Fig. 2, Table 2). No differences were observed between controls and patients within the DMN and the visual network.
Regions exhibiting differences in functional connectivity in patients with schizophrenia compared with controls and plots of the correlation analyses using the Psychotic Symptoms Rating Scale — auditory hallucination scale total score.37 Results from the second-level between-group analyses (p < 0.001, uncorrected at the voxel level, p < 0.05 corrected for spatial extent) and from the correlation analyses (p < 0.05); see also Table 2 for detailed coordinates and Z scores. AVH = auditory verbal hallucinations.
Brain regions showing connectivity differences between healthy controls and patients with schizophrenia*
Relation between functional connectivity indices and psychometric measures
Significant correlations were found between connectivity of the left ACC (ρ = −0.75, p = 0.012), the left STG (ρ = 0.71, p = 0.023) and the right MFG (ρ = 0.78, p = 0.008) with measures of overall AVH severity, as measured by the PsyRatS total score (Fig. 2). In contrast to measures of AVH severity, the PANSS-P score was positively correlated (ρ = 0.71, p = 0.021) with connectivity of the right MTG, whereas the PANSS-N score was negatively correlated (ρ = −0.72, p = 0.020) with connectivity of the right MFG (BA 8).
To investigate potential effects of antipsychotic medication in this patient sample, we additionally conducted correlation analyses using the mean voxel weights from significant between-group clusters and chlorpromazine equivalents. We found a significant correlation between chlorpromazine equivalents and connectivity of the right MTG (ρ = 0.71, p = 0.022); the other correlations were not significant.
Discussion
This study investigated the functional connectivity of multiple RSNs in patients with schizophrenia who have persistent AVHs. Two main findings emerged. First, 3 RSNs showed functional connectivity differences between patients and controls. Specifically, we found abnormal connectivity within 2 left- and right-lateralized RSNs and within an executive control network. Second, when considering overall AVH severity, we found correlations between functional connectivity and psychometric measures in the left ACC, left STG and right MFG. These regions were not significantly correlated with measures of general psychopathology, as assessed by the BPRS and the PANSS.
Considering the extant functional imaging literature, our results are in agreement with data suggesting abnormal activity of speech-related regions in patients who have AVHs. Specifically, areas of the temporal cortex, mostly left-lateralized, in conjunction with regions of the cingulate cortex have been demonstrated to exhibit increased levels of metabolism and blood flow in patients who have AVHs.2–4 We detected regions of abnormal connectivity within a left fronto-temporo-parietal system (i.e., a network associated with language processing, as described by studies of speech and language processing46 and by more recent studies of multiple RSNs27,28). Specifically, we observed disrupted connectivity of the temporal and cingulate cortices, where patients showed less connectivity in the cingulate cortex and increased connectivity in bilateral temporal regions compared with controls. The relation between measures of AVH severity and functional connectivity of the ACC and the STG further suggests a critical role for these regions in symptom generation and in the modulation of symptom intensity, which is consistent with previous reports.20,47–49 Abnormal activation and dysconnectivity of the left STG and the ACC in patients who have AVHs has been previously observed during speech-attribution tasks,14,50 providing support to the notion that a decoupling of a temporocingulate pathway could lead to speech misattribution and false auditory perceptions in hallucinating patients with schizophrenia. This pattern of functional decoupling was further confirmed by a recent study that reported a relation between AVH severity and ACC connectivity during resting-state conditions.20 The ACC has been frequently associated with directed attention, online source monitoring and cognitive control,51 and decreased connectivity of this region might parallel a failure of these processes in the context of inner speech perception. Furthermore, activation of the ACC is significantly different between self- and nonself-related stimuli,52 suggesting that the ACC could mediate the distinction between self-attributed inner speech perception and phenomena attributed to a nonself source. Of note, the disruption of temporocingulate regions in our patient sample was observed during resting-state conditions (i.e., in the absence of externally induced cognitive or linguistic stimuli), suggesting that the occurrence of internal stimulation by AVH is characterized by a similar pattern of dysfunction, as observed in studies of overt speech generation and speech attribution.50 Within the left fronto-temporo-parietal RSN, however, we did not observe AVH-related connectivity differences in the Broca region in this sample, which contrasts previous reports.53 Yet activation differences in the Broca region/inferior frontal gyrus (IFG) in patients who have AVHs or are prone to them have not been consistently reported,54–56 and this data heterogeneity may be partly explained by the diversity of the speech and cognition paradigms used in previous studies compared with resting-state or low-level demand conditions. Since we did not employ a specific activation paradigm in our study, the absence of IFG dysconnectivity could alternatively indicate that deficient generation and perception of inner speech and decreased attentional control14,57,58 rather than speech expression are among the primary disrupted functions in patients with persistent AVHs. Thus, the left STG may lie at the core of a final common pathway underlying the generation and experience of AVHs, as proposed by other researchers.3,57
Interestingly, the speech-related RSN was not the only network showing connectivity abnormalities in this study. Connectivity differences in patients who have AVHs were also detected in the right frontoparietal and the executive control network. Two features of these findings are noteworthy: first, patients showed increased functional connectivity in predominantly prefrontal regions compared with controls, and second, the pattern of dysconnectivity included mostly regions of the right hemisphere. Abnormal activation of right hemisphere regions is a frequently reported finding in patients who have AVHs, where activation changes have been most consistently reported for areas of the prefrontal and temporal cortices.45,55,59 These findings have been discussed within the context of a disrupted language lateralization in patients who have AVHs.2,60 The data provided by our study, however, suggest the involvement of an RSN distinct from the network subserving language. For instance, a right lateralized ventral attentional system comprising middle and inferior prefrontal and posterior parietal regions has been described by numerous functional neuroimaging studies of attention, including studies of intrinsic and phasic alertness61 and attentional control.62 Also, several resting-state fMRI studies have consistently reported a right frontoparietal network similar to the RSN identified in our study, possibly reflecting baseline properties of a network subserving attentional processes that persists in the absence of external events.28,63 In contrast, the executive control network, as identified by our study, has been previously referred to as corresponding to cognition paradigms of inhibitory control and affective processing.28 In conjunction with findings of abnormal connectivity in a speech-related RSN, we speculate that aberrant right middle and superior frontal connectivity within attention and executive networks could reflect increased attentional resources for salient internal events, such as the occurrence of AVHs, paralleled by inefficient top–down suppression and executive control. A similar suggestion emerged from activation studies of dichotic listening in patients who have AVHs, in whom a failure to recruit a generalized effort network4 (i.e., brain circuits necessary for adequate self-monitoring, inhibitory control and goal-directed behaviour) has been proposed to underlie AVH in addition to deficient speech-processing networks.4,6 However, since we investigated participants under resting-state conditions, the precise cognitive or affective functions of the RSN described in our study cannot be solely inferred by their spatial pattern nor fully delineated by circumscribed loci of dysfunction. Thus, the preliminary conclusions drawn from the present resting-state data set clearly need further support from multimodal neuropsychologic, task-based and resting-state fMRI studies.
Contrary to our prediction, we did not confirm DMN dysconnectivity in this patient sample. This contrasts with previous reports of disrupted DMN connectivity in patients with schizophrenia.16,25 In this regard, it is worthy to note that previous studies of the DMN in patients with schizophrenia have also reported heterogeneous findings for specific loci of DMN dysconnectivity,16,25 possibly related to clinical variation and symptom expression in a given sample. Moreover, DMN dysfunction in patients with schizophrenia has been more consistently reported within the context of cognitive tasks,25,26 where task demands and stimulus-driven interactions of task-positive (i.e., frontoparietal networks) and task-negative (i.e., DMN) systems have to be considered.17 Further research is needed to dissociate alterations of brain network connectivity during experimentally induced conditions and their interactions with baseline DMN connectivity, specifically in hallucinating patients.
Limitations
Our study has several strengths, such as the hypothesis-driven investigation of multiple functional RSNs not influenced by externally induced task-specific stimuli, the use of multivariate methods for fMRI data and the inclusion of a psychometrically well-characterized clinical population with persistent AVHs and sufficient insight into their symptoms. However, we also acknowledge several limitations. First, although our data are in agreement with the extant neuroimaging evidence suggesting abnormal frontotemporal function in patients who have AVHs, the generalizability of our results is potentially limited by the relatively small sample size. However, in contrast to most studies involving patients who have AVHs so far, we used multivariate methods, which are known to yield increased power compared with analyses conducted within the framework of the general linear model. In this regard, independent component analysis, in contrast to the general linear model, uses all scans to estimate spatially independent and temporally coherent networks, thus reducing on the first level the number of scans needed to obtain a significant response.32 We acknowledge that independent component analysis may also create a potential multiple comparison problem, since this method minimizes the mutual information among sources and may thus estimate nonorthogonal components. Currently, however, there are no available statistical tools to correct for this potential issue for fMRI data. Nevertheless, the independent component analysis method of exploratory fMRI analysis is regarded as preferable to that of, for example, principal component analysis since the spatial independence enforced upon components by (spatial) independent component analysis dictates only that their time courses should not be highly colinear, resulting in a more biologically plausible systems model than that obtained from a principal component analysis decomposition. With the latter approach, the analysis enforces orthogonality between time courses, precluding the detection of signals that partially correlate in the temporal domain.64
Second, we investigated a group of hallucinating patients only, and thus the specificity of our connectivity findings in contrast to patients who do not have AVHs must remain open at this stage of research. We found different correlations for AVH measures than for scores rating general psychopathology (BPRS) or overall positive and negative symptoms (PANSS-P and PANSS-N), suggesting that some regions could be specifically associated with the occurrence of AVHs and with AVH severity. However, the correlation results should be interpreted with caution given the limited sample size.65 It is also noteworthy that a recent study of multiple RSNs in nonhallucinating patients with schizophrenia reported a dysfunction of the DMN and the right frontoparietal network,16 suggesting that actively hallucinating and nonhallucinating patients may show different patterns of intrinsic dysconnectivity. Nevertheless, further research is needed to elucidate the specificity of RSN dysfunction in patients who have AVHs in comparison with those who do not or who have other syndromes within the schizophrenia spectrum.
Third, we included patients with chronic schizophrenia and, although our data are in agreement with results from studies of first-episode patients who have AVHs,54 patients with a longer history of schizophrenia might show different phenomenologic characteristics of AVH66 and possibly different spatial patterns of neural dysfunction. Eventually, all of the patients who participated in the present study received treatment with antipsychotic medications. Although a significant correlation was only found between connectivity of the right MTG and chlorpromazine equivalents, we cannot fully rule out potential long-term medication effects or effects of previous treatments with antipsychotics on RSN connectivity.
Conclusion
The results of the present study are suggestive of abnormal connectivity of multiple RSNs in patients with schizophrenia who have persistent, treatment-refractory AVHs. Our findings indicate that functional changes of a left-lateralized fronto-temporoparietal RSN associated with language processing and speech monitoring may underlie and modulate persistent AVHs and symptom intensity. In addition, the occurrence of hallucinatory symptoms seems to affect other RSNs, possibly interacting with attentional and executive capacity.
Acknowledgements
This work was supported by a research grant from the University of Ulm, Germany (principal investigator R.C.W.). The authors thank all participants and their families for their time and interest in this study. The authors are grateful to Miriam Ott and Petra Neumann for their assistance with data collection.
Footnotes
Competing interests: None declared from Drs. N.D. Wolf, Sambataro, Vasic, Schmid, Schönfeldt-Lecuona, Thomann and R.C. Wolf. Dr. Frasch declares having received grant support from AstraZeneca, lecture fees from Janssen and travel support from AstraZeneca, Janssen, Lilly and Pfizer; and holds stock in STADA.
Contributors: Drs. N.D. Wolf, Sambataro, R.C. Wolf, Vasic and Schönfeldt-Lecuona designed the study. Drs. N.D. Wolf, R.C. Wolf, Vasic, Frasch and Schmid acquired the data. Drs. N.D. Wolf, Sambataro, R.C. Wolf and Thomann analyzed the data. Drs. N.D. Wolf and R.C. Wolf wrote the article, which Drs. N.D. Wolf, Sambataro, R.C. Wolf, Vasic, Frasch, Schmid, Schönfeldt-Lecuona and Thomann reviewed. All authors approved publication.
- Received January 26, 2011.
- Revision received March 27, 2011.
- Revision received April 17, 2011.
- Accepted April 19, 2011.