Abstract
Background: Autonomic nervous system (ANS) dysfunction is a putative underlying mechanism for increased cardiovascular disease risk in individuals with psychiatric disorders. Previous studies suggest that this risk may be related to psychotropic medication use. In the present study we systematically reviewed and analyzed published studies of heart rate variability (HRV), measuring ANS output, to determine the effect of psychiatric illness and medication use.
Methods: We searched for studies comparing HRV in physically healthy adults with a diagnosed psychiatric disorder to controls and comparing HRV pre- and post-treatment with a psychotropic medication.
Results: In total, 140 case–control (mood, anxiety, psychosis, dependent disorders, k = 151) and 30 treatment (antidepressants, antipsychotics; k = 43) studies were included. We found that HRV was reduced in all patient groups compared to controls (Hedges g = −0.583) with a large effect for psychotic disorders (Hedges g = −0.948). Effect sizes remained highly significant for medication-free patients compared to controls across all disorders. Smaller and significant reductions in HRV were observed for specific antidepressants and antipsychotics.
Limitations: Study quality significantly moderated effect sizes in case–control analyses, underscoring the importance of assessing methodological quality when interpreting HRV findings.
Conclusion: Combined findings confirm substantial reductions in HRV across psychiatric disorders, and these effects remained significant even in medication-free individuals. Reductions in HRV may therefore represent a significant mechanism contributing to elevated cardiovascular risk in individuals with psychiatric disorders. The negative impact of specific medications on HRV suggest increased risk for cardiovascular disease in these groups, highlighting a need for treatment providers to consider modifiable cardiovascular risk factors to attenuate this risk.
Introduction
Psychiatric disorders are associated with an increased risk of all-cause mortality.1 About 60% of this increased risk is due to comorbid physical illnesses,2–4 with the most common cause of death due to cardiovascular diseases. Estimates indicate approximately a 2- to 3-fold increased risk of cardiovascular disease across a range of psychiatric disorders.5–14 The reduction in life expectancy associated with this increased risk is estimated to be greater than loss of years associated with heavy smoking alone.15
Autonomic regulation of the heart plays a key role in cardiovascular functioning.16,17 Poor cardiac autonomic regulation has been implicated in heart failure,18,19 and increased vagal input (through vagal nerve stimulation or pharmacological modulators) is protective against ventricular fibrillation20,21 and sudden cardiac death.22 Heart rate variability (HRV), the assessment of beat-to-beat variation in the heart over time, provides a reliable index of cardiac autonomic funtion.23 Healthy autonomic cardiac control is characterized by a high degree of variability in heart rate, helping to protect against myocardial infarction and other future cardiac events, while lower parasympathetic input to the sinoatrial node predicts sudden and nonsudden cardiac death.24,25 Prospective evidence indicates that reductions in HRV are strongly associated with higher incidence of first cardiac events in patients without known cardiovascular disease26 and predicts the later development of cardiovascular disease.27,28 Reductions in HRV have therefore been considered a marker for various disease states, most notably a greater risk for cardiovascular disease and all-cause mortality.29,30
A large body of evidence indicates that reductions in HRV are found in individuals with psychiatric disorders, including depression, schizophrenia, anxiety disorders and substance dependence.31–33 Influential reviews29,30 in this area have argued that reductions in HRV provide an important mechanism contributing to increased risk for sudden cardiac death in patients with these disorders.24,26,34 Other studies have highlighted the potential adverse influence of psychotropic medications, which are prescribed to treat symptoms in patients with these disorders, in reducing HRV. Some have argued that the impact of psychiatric illness on HRV is actually largely moderated by psychotropic medication use, with particular emphasis being placed on antidepressant use.35
Recently, psychiatric research has shifted to examining common biological markers across traditional diagnostic boundaries and stages of psychiatric illness severity.36,37 This approach provides a more powerful heuristic method to study biological markers that may be commonly associated with mental and physical illnesses as well as to be able to determine symptom severity. While reductions in HRV have been demonstrated in different disorders, no analysis, to our knowledge, has previously sought to examine the relative effect between and across disorders to determine what the overall and specific impact of psychiatric illness is on HRV. A greater understanding of autonomic nervous system (ANS) functioning specific or common to these disorders will also provide guidance for treatment providers when considering interventions that may reduce further risk for cardiovascular disease and other physical health concerns in these populations.
To our knowledge, there has been no systematic investigation to date of ANS functioning either across psychiatric disorders or examining the specific impact of different psychotropic medication classes. Therefore, the present study aimed to aggregate findings from studies examining HRV in patients with a psychiatric disorder without evidence of pre-existing cardiovascular diseases to examine whether HRV is reduced in individuals with psychiatric disorders compared to controls and to determine if HRV is reduced by use of psychotropic medications.
Methods
The current systematic review and meta-analysis was undertaken and reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement.38
Criteria for considering studies for this review
Types of participants
We considered studies with participants aged 18 years or older with either a primary diagnosis of an Axis I disorder (e.g., major depressive disorder, bipolar disorder, schizophrenia, generalized anxiety disorder, social anxiety disorder, panic disorder, obsessive-compulsive disorder, posttraumatic stress disorder, or substance dependence disorders) or other related disorders (e.g., dysthymic disorder, seasonal affective disorder, schizophreniform disorder, schizoaffective disorder, acute psychosis), according to formal diagnostic (DSM or ICD) criteria. Concurrent secondary psychiatric disorders in participants were not exclusion criteria; however, any study including participants with concomitant medical illnesses (e.g., cardiovascular disease, diabetes, epilepsy), elevated risk for cardiovascular disease, or including pregnant women, were excluded. Studies in which other forms of mood disturbances were examined (e.g., postpartum depression, pre-menstrual dysphoric disorder) were also excluded.
Types of studies or interventions included
Two types of studies were considered for inclusion: case–control studies comparing HRV between a psychiatric group and a healthy comparison group and double-blind, single-blind, or open-label studies comparing the effects of a psychotropic medication on HRV pre- and post-treatment. The latter included all studies of antidepressants, anti-psychotics, mood stabilizers and benzodiazepines in any oral form of administration in any psychiatric group (see Appendix 1, available at jpn.ca).
For the meta-analysis examining the impact of psychiatric illness on HRV, both medicated and medication-free (or medication-naive) participants were included. For the meta-analysis of psychotropic medication use, participants were required to be medication-free, as defined by the study authors, or medication-naive prior to participation in the study. Where a study reported data eligible for both case–control and treatment analyses, both sets of data were included.
Outcome measures
We defined HRV as any measure calculated from interbeat intervals (IBIs) derived from an electrocardiogram (ECG) or portable device (e.g., Holter, Polar heart rate monitor, pulse oximeter) measuring parasympathetic function,39 including (in hierarchical order) respiratory sinus arrhythmia (RSA); high frequency (HF; both absolute and normalized values, between 0.15 – 0.4 Hz); square root of the mean-squared difference between successive R-R intervals (RMSSD), standard deviation of all R-R intervals (SDNN), and the proportion of number of pairs of successive beat-to-beat intervals that differ by more than 50 ms (PNN50); and (4) nonlinear measures (e.g., approximate entropy; ApEn). If more than 1 of these measures was reported, only 1 was extracted according to the hierarchy specified. Both absolute and log-transformed values were included in the calculation of effect sizes. Data from both short- (approximately 5-min resting-state recordings, up to 30 min in supine, semisupine, or sitting positions) and long-term (24 h, ambulatory) recordings were included.
Quality assessment
Owing to limited tools available to assess study quality in observational and nonrandomized designs, we used a previously published measure of study quality in studies of HRV in functional somatic disorders to specifically evaluate quality of HRV methods.40 We additionally modified 1 item to assess whether drop-outs were accounted for in treatment studies (quality assessment tool available from authors on request). We assessed 3 general domains: appropriate selection of participants, appropriate quantification of HRV and appropriate control for confounding factors. A maximum score of 18 could be obtained, with a minimum score of 7 required for study inclusion.
Moderator variables
Moderators included mean age and sex distribution of the psychiatric group, diagnostic criterion used (DSM-III-R or earlier v. DSM-IV or later), duration of treatment (in days), duration of recording (short recordings were up to 30 min; long recordings were obtained from 24-hour holter ECGs), patient setting (inpatient v. outpatient), year of publication and study quality.
Search strategy
We conducted a computerized search of PubMed and EMBASE from January 1961 to February 2015 (Appendix 1). This cut-off date was selected based on the earliest known approval data from any psychotropic medication included for analysis. Phrases used for searches included (depress*, anxi*, schizo*, schizoph*, bipolar*, depend*, panic, obsess*, dysthym*, euthy*) OR (antidepress*, antipsychotic, mood stabilizer, benzo*, anticonvul*) AND (“heart rate variability,” “vagal,” “autonomic nervous system”). Only studies published in English were selected. In addition to these electronic searches, we manually examined each article’s citation list and citing articles, identified using Google Scholar, for additional studies.
Study selection and data extraction
After searches were completed, duplicate titles were removed and titles scanned. Abstracts of potentially eligible studies were then read to determine eligibility for coding. When the title and abstract could not be rejected, we obtained the full text of the article and reviewed it for inclusion using a structured data abstraction form (Appendix 1), which we piloted on a random sample of 10 studies. Reviewers (G.A.A. and D.S.Q.) assessed studies for inclusion and evaluated completeness of the data abstraction. Quality ratings were assigned by a third independent reviewer and then confirmed by G.A.A. Disagreements on any aspect of extraction or coding were solved through discussion, and consensus was achieved on all disagreements.
Data from each study (sample size, mean, standard deviation, t test value, p value) were extracted. Other data extracted included study characteristics for identification (e.g., lead author, publication year), participant characteristics (e.g., diagnostic criteria, age range, patient setting), intervention details (e.g., medication name, dose ranges, mean doses), and details about HRV measurement. Corresponding authors of papers were contacted to supplement any incomplete reporting of studies published within the last 10 years (n = 17), with a 45% response rate. For specific details regarding data extraction, see Appendix 1.
Statistical analysis
Meta-analyses were performed using Comprehensive Meta-Analysis software version 3 (Biostat Inc.), with additional analyses performed in SPSS version 21 (IBM). Effect sizes were calculated as standardized mean differences with Hedges g using the following interpretation: 0.2 = small; 0.5 = medium; 0.8 = large.41 While Hedges g offers the same interpretation as Cohen d, it also corrects for potential biases that may arise from small sample sizes, whereas Cohen d tends to overestimate the absolute value of the standardized mean difference in small samples.41 Effect size estimates were modelled using a random-effects model, which estimates a mean of a distribution of effects. Under this model, allocation of study weights is based on the inverse of the total variance, which includes both within- and between-study variance. This method is a more conservative approach, yielding a wider confidence interval (CI) when there is significant heterogeneity among effect sizes.
We conducted planned moderator and subgroup analyses in an attempt to identify significant sources of heterogeneity. Subgroup meta-analyses were conducted using a mixed- effects model. This uses a random-effects model to generate within-subgroup variance and a fixed-effects model to compare effects between subgroups. Combined effect sizes for subgroups were considered only when there were more than two studies included; effect sizes were considered significantly different from each other when the 95% CIs did not overlap with each another. We analyzed the effect of continuous moderators on the pooled effect size and the heterogeneity across studies using random-effects meta-regression.
We assessed heterogeneity using the Cochrane Q statistic, and the I2 statistic was used to evaluate the impact of heterogeneity.42 A significant Q statistic indicates dissimilar effect sizes across studies, suggesting that methodological or population sample differences might be introducing variance in findings across studies.43 We also calculated I2 to assess true heterogeneity across studies (i.e., the proportion of heterogeneity across studies not due to random error). I2 values of 25%, 50% and 75% were considered to imply small, moderate and high levels of heterogeneity, respectively.42
The sensitivity analysis was initially undertaken using forest plots to visually identify outliers (Appendix 1, Figs. S2–S5). The impact of any outliers was then addressed by the method of 1 study removed. Tests of publication bias were initially carried out by visually inspecting funnel plots of standardized mean differences against standard error and then assessed using Egger’s test of funnel plot asymmetry44 and Rosenthal’s fail-safe N.45 If publication bias was indicated (Egger regression test: p < 0.1), we used the trim and fill method for random-effects models to impute “missing studies” to redress funnel plot asymmetry,46 with adjusted pooled effect sizes and 95% CIs reported after the addition of potential missing studies.
Results
Description of included studies
The search of electronic databases resulted in 9579 studies (Appendix 1, Fig. S1). Duplicates were removed, titles and abstracts scanned, potential articles read and evaluated against inclusion criteria and authors contacted for additional data when necessary. In total, 140 case-control47–186 and 30 treatment76,83,93,110,116,129,138,141,155,175,187–206 studies were eligible for inclusion in the quantitative synthesis (151 case–control and 43 effect sizes for analysis); 10 of the studies were included in both the case–control and treatment analyses.76,83,93,110,116,129,138,141,155,175 Four subgroups of disorders (mood, anxiety-related, psychotic and substance dependence disorders) and 2 classes of psychotropic medications (anti-depressants and antipsychotics) were included.
Table 1 reports study characteristics for case–control and treatment studies, separated by diagnostic group or type of psychotropic medication, respectively (see Appendix 1, Tables S1 and S2, for individual study characteristics). In case–control studies, there were significantly higher proportions of men in the psychotic and substance dependence disorder than in the mood and anxiety disorder subgroups. Patients in the mood and substance dependence groups were also significantly older. In the treatment studies, patients in antidepressant studies were significantly older, whereas those in antipsychotic studies included a significantly higher proportion of men.
Characteristics of included studies
Included studies reported a mixture of linear (frequency and time domain) and nonlinear outcomes. Of the 160 included studies, 64% reported a frequency measure (absolute or normalized HF), with the remainder reporting RSA (9%), time domain (RMSSD, SDNN; 20%), or nonlinear (7%) measures. The majority of studies (90%) reported HRV outcomes from short-term (< 1 h) recordings, with the remaining studies using ambulatory recordings taken over 24 hours. The average duration of treatment in antidepressant studies (mean 33.857 ± 25.730 d) was not significantly different from the duration of treatment in antipsychotic studies (mean 33.733 ± 24.271 d, p = 0.99).
Quantitative analyses
Effects of psychiatric disorder on HRV
Overall, HRV was significantly reduced in patients compared to controls (Hedges g = −0.582, 95% CI −0.664 to −0.501, p < 0.001; Table 2). Both heterogeneity (Q = 645.563, p < 0.001) and Egger’s test (p < 0.001, 2-tailed) were significant, indicating significant variability in effect sizes among studies and significant publication bias, respectively. The level of true heterogeneity was large (I2 = 76.55%), meaning that only about 25% of the heterogeneity was due to sampling error. For publication bias, visual inspection of the funnel plot confirmed asymmetry; in the trim and fill analysis, 29 studies were imputed, resulting in an adjusted larger point estimate (Hedges g = −0.746, 95% CI −0.835 to −0.656).
Meta-analytic results of HRV in patients compared to controls across psychiatric subgroups and current medication use
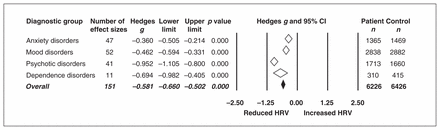
The planned subgroup analysis revealed that while all psychiatric disorder groups exhibited significant reductions in HRV compared to controls, the largest effect was observed in patients with psychotic disorders compared to controls (n = 41, Hedges g = −0.952, 95% CI −1.105 to −0.800, p < 0.001), associated with a large effect size. A lack of overlapping CIs indicated that this effect was significantly larger than overall effect size estimates for mood or anxiety disorder subgroups (Fig. 1).
Summary effect estimates comparing significant reductions in heart rate variability (HRV) in psychiatric disorder subgroups compared to healthy controls. Open diamonds depict summary effect sizes of each subgroup; filled diamond reflects the overall summary effect across groups. Diamond width indicates the 95% confidence interval (CI) for each summary effect size.
The subgroup analysis comparing medication use indicated that nonmedicated patients overall exhibited a smaller significant reduction in HRV (Hedges g = −0.471, 95% CI −0.577 to −0.365, p < 0.001) compared to a moderate effect size in medicated patients (Hedges g = −0.734, 95% CI −0.859 to −0.609, p < 0.001), with significant heterogeneity (Q = 9.403, p = 0.002) and lack of overlapping CIs indicating significant differences in these effect sizes (Table 2). When examining diagnostic subgroups, this moderating effect of medication use on HRV appeared to remain only for patients with mood disorders (Appendix 1, Figs. S2–S5).
Diagnostic subgroup analysis was then repeated with only studies reporting inclusion of nonmedicated patients (medication-naive or medication-free) compared to controls. The overall effect of HRV remained highly significant (n = 82, Hedges g = −0.468, 95% CI −0.565 to −0.371, p < 0.001), with evidence of heterogeneity of effect sizes (Q = 17.851, p = 0.001). The magnitude of each effect size remained the same for each subgroup, with small effect sizes noted in anxiety (n = 30, Hedges g = −0.335, 95% CI −0.502 to −0.168, p < 0.001) and mood disorders (n = 29, Hedges g = −0.349, 95% CI −0.505 to −0.193, p < 0.001), a moderate effect size for substance dependence disorders (n = 4, Hedges g = −0.612, 95% CI −1.060 to −0.165, p = 0.007) and a large effect size for psychotic disorders (n = 19, Hedges g = −0.840, 95% CI −1.043 to −0.636, p < 0.001). Egger’s test of publication bias remained significant (p = 0.009), and the trim and fill analysis imputed 12 studies to the left of the mean; this increased the point estimate (Hedges g = −0.578, 95% CI −0.680 to −0.476).
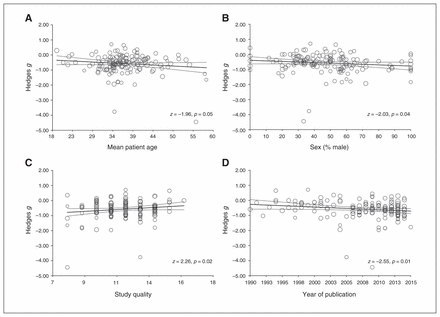
Moderator analyses revealed no significant differences due to different HRV outcome measures (RSA, frequency, duration, or nonlinear domains) or duration of recording (short or long). Significantly different effect sizes (heterogeneity p < 0.001) were found for studies using inpatients (n = 62, Hedges g = −0.710, 95% CI −0.826 to −0.595, p < 0.001) compared to outpatients (n = 73, Hedges g = −0.407, 95% CI −0.513 to −0.301, p < 0.001). Similarly, significant differences were observed for studies that used earlier diagnostic criteria (ICD-9, DSM-III-R or earlier; n = 30, Hedges g = −0.367, 95% CI −0.549 to −0.185, p < 0.001) compared to more recent versions (ICD-10, DSM-IV or later; n = 121, Hedges g = −0.636, 95% CI −0.727 to −0.545, p < 0.001). Meta-regression analyses revealed significant regressions of Hedges g on age (p = 0.05), sex (p = 0.043), study quality (p = 0.024) and year of publication (p = 0.011). This suggested that more recent and lower-quality case–control studies as well as studies with an older mean age of participants or greater proportion of men were significantly associated with larger effect sizes (Fig. 2).
Results of meta-regression analyses in case–control studies examining effects of continuous moderators on the effect sizes of (A) mean patient age, (B) the proportion of men in the patient group, (C) independent study quality ratings and (D) year of publication. Studies are indicated by open circles, with size indicating relative weight.
Effects of psychotropic medications on HRV
The overall analysis indicated small but significant reductions in HRV after use of psychotropic medications (Hedges g = −0.206, 95% CI −0.353 to −0.059, p = 0.006; Table 3). Tests of heterogeneity (Q = 216.748, I2 = 80.623, p < 0.001) were significant, with only about 20% of the true heterogeneity attributable to sampling error; Egger’s test of publication bias was not significant.
Meta-analytic results of the effects of psychotropic medications on HRV
Subgroup analyses revealed a small but significant reduction in HRV in the mood disorder group (Hedges g = −0.221, 95% CI −0.436 to −0.005, p = 0.045), but not in the anxiety and psychotic disorder groups (Table 3). This suggested that any effects of antidepressant use on HRV may be larger in individuals with mood disorders. Direct comparison of type of medications indicated significant reductions in HRV for both antidepressant (Hedges g = −0.206, 95% CI −0.400 to −0.012, p = 0.037) and antipsychotic use (Hedges g = −0.215, 95% CI −0.421 to −0.009, p = 0.041).
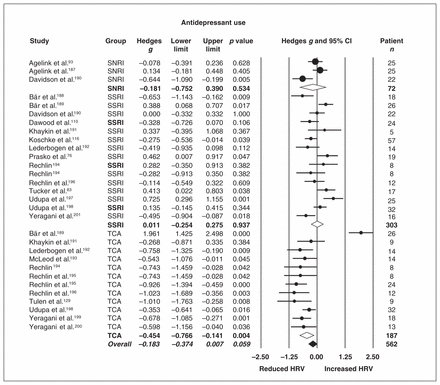
Subgroup analyses comparing types of antidepressants revealed significantly reduced HRV after tricyclic antidepressant (TCA) administration (p = 0.004; Fig. 3), which was associated with a small to moderate effect size, compared to other anti-depressants (selective serotonin reuptake inhibitors [SSRIs] and serotonin–norepinephrine reuptake inhibitors [SNRIs]).
Effects of antidepressant use on heart rate variability (HRV) in individuals with a mood or anxiety disorder. Removal of the outlier in the tricyclic antidepressant (TCA) group189 shifted the effect size of the TCA subgroup (Hedges g = −0.666, 95% confidence interval [CI] −0.900 to −0.432, p < 0.001) and the overall effect size (Hedges g = −0.231, 95% CI −0.364 to −0.097, p = 0.001). Point estimates are depicted by filled circles, with sizes indicating the relative weight of each study’s effect size estimate to the analysis. Open diamonds depict summary effects sizes of each subgroup; the filled diamond reflects the overall summary effect across groups. Line and diamond width indicate 95% CIs. SNRI = serotonin-norepinephrine reuptake inhibitor antidepressant; SSRI = selective serotonin reuptake inhibitor antidepressant.
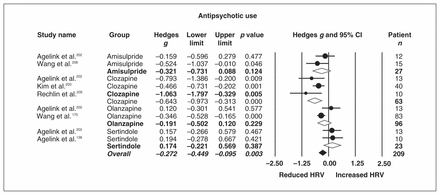
To explore significant reductions in HRV due to antipsychotic use, we conducted a subgroup analysis including all antipsychotics where there was more than 1 study examining the effects of that antipsychotic’s administration on HRV. This left 4 antipsychotics for comparison (amisulpride, clozapine, olanzapine and sertindole — all atypical anti-psychotics). While the overall summary effect size for anti-psychotic use remained significant (Hedges g = −0.272, 95% CI −0.449 to −0.095, p = 0.003), only clozapine use had a significant detrimental effect on HRV (Hedges g = −0.643, 95% CI −0.973 to −0.313, p < 0.001), a point estimate associated with a medium effect (Fig. 4).
Effects of antipsychotic use on heart rate variability (HRV) in individuals with schizophrenia. Point estimates are depicted by closed circles, with size indicating the relative weight of each study’s effect size estimate to the analysis. Open diamonds depict summary effect sizes of each subgroup; filled diamond reflects the overall summary effect across groups. Line and diamond width indicate 95% confidence intervals (CIs).
Moderator analyses revealed no significant differences in the overall effect estimates of psychotropic treatments due to type of HRV measure, patient setting, diagnostic criteria used, recording duration, sex, mean age, or study quality (all p > 0.05). Tests of publication bias were nonsignificant across subgroup analyses, with sensitivity analyses confirming relative robustness of effect size estimates (Table 3).
Discussion
The present meta-analysis investigated ANS functioning in patients with psychiatric disorders and the potential moderating effects of psychotropic medications. Results suggested 2 main findings: ANS dysfunction is a characteristic feature in otherwise healthy patients with a major psychiatric disorder, with the largest effects observed in patients with psychotic disorders; and psychotropic medications have a small impact in further reducing HRV in these populations, specifically associated with TCA and clozapine use. It can therefore be concluded that many psychotropic medications do not negatively impact ANS functioning, an important consideration given reduced HRV and elevated risk for cardiovascular disease in these populations. However, results suggest increased vigilance and monitoring in the use of these specific psychotropics in patients. Importantly, the results highlight the critical need for treatment providers to additionally address modifiable factors for patients that may contribute to reductions in cardiovascular disease risk, such as exercise, diet and smoking cessation.
ANS functioning in patients with psychiatric disorders
The overall summary effect indicated reduced HRV in patients compared to controls with a moderate effect size. The robustness of this effect across disorders suggested that ANS dysfunction, with the associated cardiovascular risk, is evident across psychiatric disorders. The strength of these summary effects also suggests particular elevated risk for psychotic disorders. Complementary to these findings, patients with a psychiatric disorder have significantly increased rates of modifiable lifestyle risk factors for cardiovascular disease, including obesity, diabetes, hypertension, increased alcohol use and smoking as well as higher rates of major physical health conditions,4 all of which may contribute to further reducing HRV in these populations. While individuals with a psychiatric illness are more likely to experience sudden death,207,208 they are also less likely to seek appropriate health care during acute cardiac episodes.209 Taken together, these findings suggest that increased monitoring of known risk factors in these populations is of key importance for treatment providers.
Effect sizes observed in the present study were larger than previously observed in other meta-analyses, specifically those examining depressive,31,210 anxiety211,212 and substance dependence32 disorders. This difference is likely owing to the larger number of studies retrieved owing to the rapidly increasing number of publications within this field in the last 5 years, allowing for more precise summary effect size estimates. By pooling patients within and across traditional disorder groupings, these results provide evidence to suggest that ANS dysfunction may be an appropriate physiological marker of cardiovascular risk for patients with psychiatric disorders. As psychiatric research (e.g., National Inistitute of Mental Health’s Research Domain Criteria [RDoC] project213) moves towards examining shared underlying biological, cognitive and neural abnormalities across psychiatric disorders,36,37 the results of the present study highlight the importance of examining common risk factors for mental and physical health disorders. Combined with cumulative evidence from cardiovascular and epidemiological studies highlighting less adaptive ANS functioning as a prognostic marker for cardiovascular risk,26,214 such findings strongly suggest the importance of more targeted and adequate prevention strategies for reducing other modifiable cardiovascular risk factors in these psychiatric populations.
Effects of psychotropic medication use on HRV
Overall, we observed a small effect of medication use driven by specific effects of TCA and clozapine use. Most anti-depressants, even those with high antimuscarinic potency, such as paroxetine, are safe to use in high-risk cardiac populations, with few cardiac side-effects.215 However, the increased risk of cardiac events in TCAs has previously been noted.216 They are associated with increased heart rate, orthostatic hypotension, slow cardiac conduction, increased risk of arrhythmias, and a 2-fold relative risk of myocardial infarction.216 Although the potential adverse effect of antidepressants on HRV have been debated,31,35,210,217–220 these previously reported negative effects may be due to the inclusion of depressed participants who were already at an elevated risk for cardiovascular disease.35 As the present meta-analysis excluded studies of individuals with known medical conditions that may influence HRV, the moderating effect of medication use in the case–control analysis is of particular interest and suggests that TCA use may exacerbate HRV reductions in patients with mood disorders. However, we found small but highly significant effect sizes for medication-free patients, suggesting that effects cannot be solely attributable to medication use or other lifestyle factors.
Despite the relatively smaller number of studies retrieved investigating antipsychotic use, the present findings provide preliminary evidence to highlight a particular negative effect of clozapine use in reducing HRV. These effects are in line with those of other systematic reviews highlighting greater risk for cardiometabolic disorders through increased weight gain and changes in cardiac function after prolonged periods of antipsychotic use.221 There are a number of direct and indirect mechanisms by which second-generation anti-psychotic drugs like clozapine may alter HRV, particularly by direct blockade of adrenergic and cholinergic receptors on the heart and blood vessels, but also indirectly though baroreflex receptors and through known metabolic side-effects, such as weight gain.222 However, it has also been argued by some that antipsychotic-induced reductions in HRV indicate autonomic imbalance,202,223 reflecting greater sympathetic and reduced vagal influence over the heart. While increased cardiovascular mortality in psychotic disorders has been attributed to modifiable lifestyle factors,209 such as higher rates of smoking, lack of exercise and substance misuse, the present meta-analysis suggests that autonomic dysfunction exists even in medication-free individuals and that some psychotropics may further exacerbate cardiac risk in these individuals.
Limitations and future directions
The purpose of the present study was to examine the combined effect of psychiatric disorders on HRV. We did not, therefore, investigate the relative strength of effect sizes within single disorders. Considerable comorbidity and overlap exists between many disorders, particularly between mood and anxiety disorders and between mood and psychotic disorders.224,225 A more important question for future research may be what impact age and earlier manifestions of the disorder have on changes in HRV over time, including first-episode and prodromal periods, as symptoms change or worsen.65,226,227 Additionally, the inclusion of cross-sectional studies in the case–control analysis does not address the question of whether reduced vagal control in these populations is part of a potential mechanism placing these individuals at a greater risk for cardiovascular disease, or whether reduced HRV in these groups compared to controls is actually a downstream effect of altered neural pathways that control autonomic regulation of the heart.228
Our search strategy included other classes of psychotropic medications (e.g., mood stabilizers, benzodiazepines) and studies of medication effects in individuals with substance dependence. However, these searches did not yield any study meeting our inclusion criteria. Given that these other classes of medications are widely used within these psychiatric groups and that psychotropic medications are often used adjunctively in the treatment of substance dependence in practice, it is of considerable interest for future research to examine the effects that these medications may have on HRV.
Sex and age were small but significant moderators of effect size in the case-control analysis. As parasympathetic activity is generally greater in women229 and associated with lower cardiovascular risk and greater life expectancy, further investigation into the effects of sex on reductions in HRV and changes in cardiac risk across disorders would be of substantial interest. While age is robustly associated with HRV, with older age associated with declining HRV,230 the magnitude of this effect was very small in the case–control meta-regression and may suggest both an effect of age and chronicity of psychiatric illness on HRV.
Study quality and year of publication were also significant moderators for case–control studies, suggesting that poorer quality studies tended to be associated with larger effect sizes, and the magnitude of effect sizes between cases and controls tended to increase over time, with inceasing numbers of studies being published. Such findings highlight the critical importance of thoroughly assessing methodology in HRV studies when interpreting the magnitude of these effects. Such methodological variations that may influence estimates of vagal control include the duration of prerecording rest periods, breathing rate (free or paced), recording conditions (supine, semisupine, or sitting resting conditions or ambulatory recordings), circadian rhythym (including time of day when recording), specific outcome measures (frequency, time and nonlinear domains), inconsistent reporting of frequency bandwidths and normalization methods for spectral measures, and limited reporting of test–retest reliability in repeated-measure designs.231,232 We controlled for many of these methodological differences in our study selection and assessed the risk of bias from variation in these measures in our quality assessment. Given the association between physical activity and HRV233,234 and the higher rates of sedentary activity in psychiatric populations associated with greater risk for cardiovascular disease,235,236 measurement of habitual physical activity levels and body mass index is important. Reporting of other factors known to modify HRV, such as caffeine,237 nicotine238 and alcohol intake,239 was also inconsistent among studies and was therefore unable to be collated for moderator analysis. However, given known greater prevalence of higher BMI and smoking rates in these populations, it is of critical importance for research in this area to appropriately report and control for these known factors.
Conclusion
The present comprehensive meta-analysis demonstrated a robust association of reductions in ANS functioning in patients with psychiatric disorders compared to controls, irrespective of psychotropic medication use. Reductions in HRV were most pronounced in otherwise physically healthy patients with psychotic disorders, although all disorders exhibited small to moderate reductions in HRV compared to controls. Current psychotropic medication moderated these findings only in patients with mood disorders, and specific use of TCAs and clozapine was associated with significantly reduced HRV. The findings from this systematic review and meta-analysis provide support for treatment providers to increase vigilance when using these psychotropics in patient groups with compromised vagal functioning. The overall findings also suggest that reduced HRV may be a putative mechanism underlying elevated risk for cardiovascular disease in patients with psychiatric disorders, with the strongest evidence for this in those with psychotic disorders. Therefore, it is highly recommended that health care practitioners monitor and emphasize treatment of modifiable risk factors associated with cardiovascular disease for individuals presenting with elevated psychiatric symptoms early in illness progression.
Acknowledgements
The authors thank Melissa Warshavsky for her assistance in data retrieval and quality assessment ratings, Anne Masi for statistical advice and Yu Sun Bin for critical assessment of the final manuscript. This work was supported by an Australian Research Council Linkage Grant (LP110100513) and a National Health and Medical Research Council Career Development Fellowship (1061922) to A.J. Guastella and an Australia Fellowship (494914) to I.B. Hickie. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Footnotes
Contributors: All authors designed the study. G. Alvares and D. Quintana acquired the data, which all authors analyzed. G. Alvares wrote the article, which all authors reviewed and approved for publication.
Competing interests: I.B. Hickie is a Senior Principal Research Fellow of the Australian National Health & Medical Research Council (1046899). He is the executive director of the Brain and Mind Research Institute (BMRI) at the University of Sydney, which operates 2 early-intervention youth services under contract to headspace. He is a commissioner of the Australian National Mental Health commission and was previously the CEO of beyondblue: the national depression initiative and a director of headspace: the national youth mental health foundation until January 2012. Previously, he has led a range of community-based and pharmaceutical industry-supported depression awareness and education and training programs. He has led depression and other mental health research service evaluation or investigator-initiated research projects that have been supported by a variety of pharmaceutical partners. Current investigator-initiated studies are supported by Servier (manufacturers of agomelatine) and Pfizer. He has received honoraria for his contributions to professional educational seminars related to depression, youth mental health and circadian rhythms research. He has received travel support from Servier to attend scientific meetings related specifically to circadian-rhythm disorders. No other competing interests declared.
- Received August 6, 2014.
- Revision received June 6, 2015.
- Accepted June 24, 2015.