Cytochrome P450 monooxygenase genes in the wild silkworm, Bombyx mandarina
- Published
- Accepted
- Received
- Academic Editor
- Kenta Nakai
- Subject Areas
- Agricultural Science, Bioinformatics, Entomology, Genomics, Zoology
- Keywords
- Bombyx mandarina, Wild silkworm, Cytochrome P450, Gene family
- Copyright
- © 2021 Wan et al.
- Licence
- This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, reproduction and adaptation in any medium and for any purpose provided that it is properly attributed. For attribution, the original author(s), title, publication source (PeerJ) and either DOI or URL of the article must be cited.
- Cite this article
- 2021. Cytochrome P450 monooxygenase genes in the wild silkworm, Bombyx mandarina. PeerJ 9:e10818 https://doi.org/10.7717/peerj.10818
Abstract
Wild (Bombyx mandarina) and domestic silkworms (B. mori) are good models for investigating insect domestication, as 5000 years of artificial breeding and selection have resulted in significant differences between B. mandarina and B. mori. In this study, we improved the genome assemblies to the chromosome level and updated the protein-coding gene annotations for B. mandarina. Based on this updated genome, we identified 68 cytochrome P450 genes in B. mandarina. The cytochrome P450 repository in B. mandarina is smaller than in B. mori. Certain currently unknown key genes, rather than gene number, are critical for insecticide resistance in B. mandarina, which shows greater resistance to insecticides than B. mori. Based on the physical maps of B. mandarina, we located 66 cytochrome P450s on 18 different chromosomes, and 27 of the cytochrome P450 genes were concentrated into seven clusters. KEGG enrichment analysis of the P450 genes revealed the involvement of cytochrome P450 genes in hormone biosynthesis. Analyses of the silk gland transcriptome identified candidate cytochrome P450 genes (CYP306A) involved in ecdysteroidogenesis and insecticide metabolism in B. mandarina.
Introduction
The domestic silkworm, Bombyx mori, is a model insect often used to study physiology, biochemistry, developmental biology, neurobiology, and pathology (Kawamoto et al., 2019). It has been reared for more than 5,000 years for silk production (Fang et al., 2015), and it is now used for commercial production of medically and industrially important biomaterials based on genetic engineering. The wild silkworm (B. mandarina), the direct ancestor of B. mori, is a valuable gene pool resource that can be exploited and utilized. B. mandarina provides an important basic material to study the origin and differentiation of silkworms and is significant for B. mori breeding and the establishment and application of a gene pool for special traits.
A draft sequence of the B. mori genome was first reported by Chinese and Japanese groups (Xia et al., 2004), and the annotation of both WGS data sets with 8.48x sequence coverage was completed in 2008 (International Silkworm Genome C, 2008). Since then, many transcriptomic and evolutionary studies have investigated important bio-systems differing between the domestic and wild silkworms based on the genome sequence. Kawamoto et al. (2019) performed hybrid assembly of B. mori based on 140 × deep sequencing of long (PacBio) and short (Illumina) reads and annotated the new genome with more RNASeq and protein data, generating higher quality genome assemblies and more accurate gene models. To reconstruct the domestication processes and to identify selective sweeps among B. mori strains, Xiang et al. (2018) generated a draft assembly for B. mandarina using a classic shotgun approach based on Illumina sequencing platforms. These high-throughput datasets provide a more comprehensive way to study the similarities and differences between B. mori and B. mandarina at the whole genome level.
The cytochrome P450 monooxygenases (P450s) genes are a large and complex gene superfamily of heme-thiolate proteins. They are found in most organisms from prokaryotes to eukaryotes (Nelson, 1999). The cytochrome P450 genes are an ancient enzymatic system, and all of the current cytochrome P450s may have descended from a common ancestral gene (Ai et al., 2011; Yu et al., 2015). P450 genes are responsible for the oxidative metabolism of structurally diverse endogenous and exogenous compounds (Nebert & Gonzalez, 1987). In insects, the cytochrome P450 enzymes catalyze the metabolism of physiologically important endogenous compounds, including hormones and pheromones at juvenile and molting stages, and they are well known for their detoxification in pesticides (Scott, 1999; Scott, Liu & Wen, 1998). For this reason, many cytochrome P450s have been studied in insects, including Drosophila melanogaster, Anopheles gambiae, Aedes aegypti, Tribolium castaneum, Apis mellifera, and B. mori.
Based on the silkworm draft genome, Ai et al. (2011) identified 84 CYP-related sequences, which were classified into 26 families and 47 subfamilies according to the standard nomenclature. However, Kawamoto et al. (2019) identified a total of 83 cytochrome P450 genes using a high-quality genome assembly. Considering the important role of B. mandarina for B. mori breeding and the important roles of cytochrome P450s in silkworms, it is interesting to explore the cytochrome P450 genes in B. mandarina and compare them to those in B. mori. In this study, the cytochrome P450 genes in B. mandarina were identified and a comprehensive genome-wide comparative analysis of the cytochrome P450 genes in B. mandarina and B. mori was performed. We linked the B. mandarina scaffold sequences generated by Xiang et al. (2018) into pseudo-molecules using their syntenic to chromosomes in the B. mori. This study illuminates the functional diversities and the evolutionary mechanisms and significance of cytochrome P450s in both B. mori and B. mandarina.
Materials & Methods
Genome and annotation data resources
Genomic and annotation data for B. mori were downloaded from SilkBase (http://silkbase.ab.a.u-tokyo.ac.jp/cgi-bin/download.cgi). This genome was published in 2019 and based on 140 × deep sequencing of long (PacBio) and short (Illumina) reads. The new genome was annotated with more RNASeq and protein data, generating higher quality genome assemblies and more accurate gene models than the previous version (Kawamoto et al., 2019). Genomic and annotation data for B. mandarina were downloaded from the NCBI RefSeq (accession: GCF_003987935.1, published in 2019) and assembled using a classic whole genome shotgun approach, based on Illumina sequencing platforms (Xiang et al., 2018). Genome and annotation datasets for other insect species are shown in Table S1.
Improving the quality of the B. mandarina genome assembly
To build chromosomes from genome contigs or scaffolds, we used an alignment-based approach. We used Lastz (Harris, 2007) to perform the whole genome alignment between B. mandarina (Xiang et al., 2018) and B. mori (Kawamoto et al., 2019). Then, using the syntenic alignments between B. mandarina contigs and scaffolds and B. mori chromosomes, we recorded the strands and turns of B. mandarina contigs and scaffolds relative to the B. mori chromosomes. Finally, the wild and domestic contigs and scaffolds were linked into pseudo-molecules according to the strands and turns information (Table S2).
Updating the gene annotation of B. mandarina
The original B. mandarina NCBI RefSeq protein-coding gene models were generated by the automated NCBI Eukaryotic Genome Annotation Pipeline (https://www.ncbi.nlm.nih.gov/genome/annotation_euk/Bombyx_mandarina/100/). We first removed the low-quality protein-coding genes with coding sequences (CDS) shorter than 150 bp and with stop codons in the CDSs. For the protein-coding gene models with alternative splicing, we only kept the longest CDS for each gene to generate a clean RefSeq annotation file in GFF3 format.
Transcriptome sequencing raw data were downloaded from previous studies (Table S3). To obtain high-quality clean reads, the raw sequencing reads were filtered using Trimmomatic software (Bolger, Lohse & Usadel, 2014) with the following steps. First, reads with adaptor sequences were removed. Then, reads containing more than 20% of low-quality bases (Q < 20) or containing more than 3% of ambiguous ‘N’ were discarded. The reads were also trimmed where the four-bases-window had an average quality lower than 20. After these filtering steps, the clean reads from each sample were aligned to the updated genome assemblies using HiSAT2 (version 2.0.4) (Kim, Langmead & Salzberg, 2015), which generated BAM files for downstream analyses.
The transcripts in each sample were assembled and merged using StringTie (version 1.3.1c) (Pertea et al., 2015) and the BAM files were generated by the HiSAT2 (Kim, Langmead & Salzberg, 2015). If none of the transcripts in a gene model had overlaps with the RefSeq protein-coding gene models, the transcripts in this gene were subjected to TransDecoder (version r20140704) (Haas, 2014) to predict the potential coding sequences (CDSs). This gene model was defined as a novel protein-coding gene model if it met the following requirements: (1) the CDSs had at least 50 codons; and (2) the CDSs had start- and stop-codons. We only retained the longest CDS for each novel protein-coding gene model.
To determine the functional annotation of the protein-coding gene models, a BLASTP (Camacho et al., 2009) search with an E-value ≤ 1e−5 was performed against protein databases, including NR (non-redundant protein sequences in NCBI) and SwissProt (Boeckmann et al., 2003). KEGG annotation that maps the B. mandarina cytochrome P450 genes to possible KO numbers and map numbers was fetched from KOBAS (version 3.0) (Xie et al., 2011) results. The domains and GO terms of each gene model were predicted by InterProScan (Quevillon et al., 2005) against public protein databases, including ProDom (Bru et al., 2005), PRINTS (Attwood et al., 1994), Pfam (Bateman et al., 2004), SMART (Ponting et al., 1999), PANTHER (Mi et al., 2005), PROSITE (Hulo et al., 2006), and TIGR (Haft, Selengut & White, 2003).
Synteny analysis of B. mandarina and B. mori
To explore the collinearity of B. mandarina and B.mori, the updated B. mandarina proteome was blasted against to the B. mori proteome, and the MCScanX (Wang et al., 2012) was used to detect syntenic genes and blocks (regions with at least eight collinear genes). The collinearity of genes between B. mandarina and B. mori was visualized using Circos (Krzywinski et al., 2009).
In silico identification of cytochrome P450 genes in B. mandarina and other insects
The cytochrome P450 genes of B. mandarina were identified as following steps. First, we collected total 84 cytochrome P450 genes in B. mori, which included 82 cytochrome P450 genes from Kawamoto et al. (2019) and another two cytochrome P450 genes identified by Ai et al. but missing in Kawamoto et al. (Ai et al., 2011). The proteins and CDSs were extracted for downstream analyses. We also downloaded 91 P450 proteins from Drosophila melanogaster (http://drnelson.uthsc.edu/cytochromeP450.html), and then the cytochrome P450 proteins from D. melanogaster and B. mor i were combined to form a reference cytochrome P450 protein database. Second, the B. mandarina proteome was searched against the reference cytochrome P450 protein database using the BLASTP algorithm (Camacho et al., 2009), with an e-value cut-off of 1e−5. For each B. mandarina protein, the top hit to the reference cytochrome P450 protein database was remained, and the B. mandarina proteins with hits to the reference cytochrome P450 proteins were the candidate cytochrome P450 proteins. Third, the domains of the B. mandarina cytochrome P450 candidates were annotated by the iprscan (version 5) (Quevillon et al., 2005) using the Pfam database (http://pfam.sanger.ac.uk/) (Bateman et al., 2004), and the candidate was identified as the cytochrome P450 gene if it had the PF00067 domain (cytochrome P450). Additional TBLASTN (Camacho et al., 2009)searches against the B. mandarina genomic assemblies followed by gene structure refinement using GeneWise (Birney, Clamp & Durbin, 2004) were also performed with the reference cytochrome P450 protein database to avoid missing cytochrome P450-related genes. We further manually corrected annotation for three genes (XP_028036989.1, XP_028036546.1 and XP_028040841.1) that were mis-predicted to generate artificial fusion genes by the NCBI Eukaryotic Genome Annotation Pipeline, and for these loci, we applied these criteria to define genuine protein-coding genes: (1) insertions, deletions or frameshifts were not allowed when they were compared to their homologous protein; (2) start codon and stop codon were compulsive, and (3) they had at least 50 amino acids.
The cytochrome P450 genes in other insect species (Table S1) were identified using the same pipeline. The exons and domains positions of silkworm cytochrome P450 genes were extracted from GFF3 file and Iprscan results, respectively, and then were plotted using the iTOL (interactive Tree Of Life, https://itol.embl.de/) (Letunic & Bork, 2019). The chromosomal locations for cytochrome P450 genes were plotted using karyoploteR (Gel & Serra, 2017).
Phylogenetic analyses and classification of B. mandarina cytochrome P450 genes
The cytochrome P450 proteins were aligned using MUSCLE v3.8.31 (Edgar, 2004) with default parameters. RAxML (version 8.2.7) (Stamatakis, 2014) was used to generate maximum likelihood with PROTGAMMALGX model and 100 bootstraps. Trees were plotted by the iTOL (https://itol.embl.de/) (Letunic & Bork, 2019). The phylogenetic tree between B. mandarina and B. mori and the standard nomenclature of B. mori were used to classify and name the B. mandarina cytochrome P450 genes (Ai et al., 2011; Kawamoto et al., 2019).
RNA-Seq expression analysis of cytochrome P450 genes
The gene expression levels of each sample were quantified using HTSeq (version 0.9.1) (Anders, Pyl & Huber, 2014) and the BAM files generated by the HiSAT2 (Kim, Langmead & Salzberg, 2015). Then, the DEGs between different sample pairs were detected via the edgeR software package (Robinson, McCarthy & Smyth, 2010) of the R language. The P-values were corrected for false discovery rate (FDR) using multiple tests. Differentially expressed genes (DEGs) met the following criteria: FDR (adjusted P) <0.05 and —log2 fold change (FC)—>0.5.
GO and KEGG enrichment analysis
Significantly over-represented GO terms among the B. mandarina cytochrome P450 genes were identified using the topGO package (Alexa & Rahnenfuhrer, 2010) in R programming language ( https://www.r-project.org/). The enrichment of KEGG pathways was conducted with Fisher’s exact test using R scripts. The significantly over-represented GO terms and KEGG maps were identified with adjusted p-values ≤ 0.05.
Results
Improvement of genome assembly to the chromosome level
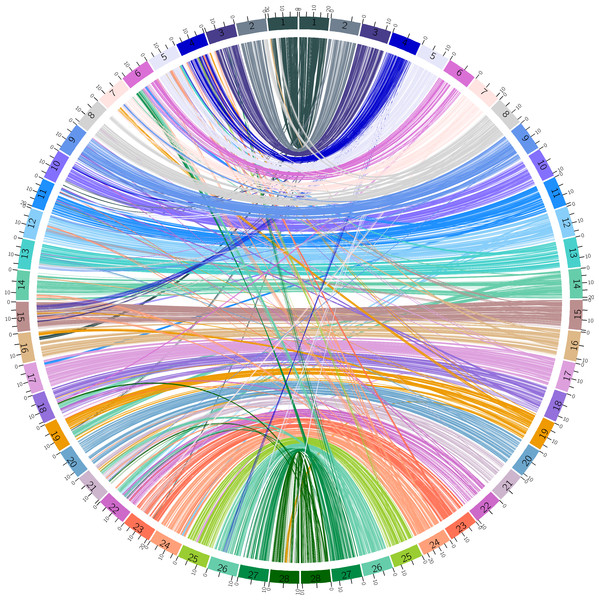
The draft assembly for B. mandarina described by Xiang et al. was fragmental and low quality (Xiang et al., 2018). There are a total of 3,105 scaffolds with a total length of 398,588,931 bp (395,983,407 bp without gaps). The scaffold and contig N50 were 2,789,315 bp and 29,637 bp, respectively. The minimum sequence length was 500 bp, and the maximum sequence length was 14,129,094 bp. To improve the quality of genome assembly for B. mandarina, we implemented a reference-assisted approach to build chromosomes from genome contigs or scaffolds published by Xiang et al. (2018). A total of 337 scaffolds were linked into 28 chromosomes, and the total length of these chromosomes reached 98.35% (392,034,257 bp) of the raw assemblies published by Xiang et al. . (Xiang et al., 2018) (Table S2). The shortest chromosome was chr2 (7,258,460 bp), while the longest one was chr24 (22,989,029 bp). Chr12 had the smallest size variation among B. mandarina and B. mori, while chromosome 4 had the largest size variation. To study the synteny of the two silkworms, the collinearity of genes between B. mandarina and B. mori was visualized using Circos (Fig. 1).
Figure 1: Synteny between B. mandarina and B. mori.
The left 28 chromosomes are from the B. mandarina genome and the right 28 chromosomes are from the B. mori genome. Echo chromosome number is marked on the karyotype bar on the circle, and ticks on each bar are the physical positions (unit: Mbp). Links are the homologous gene pairs identified by MCScanX and colored using the B. mori chromosome color scheme.Identification of novel protein-coding genes and gene function annotation
To obtain more protein-coding gene models in the B. mandarina genome, we updated the gene annotation using StringTie-TransDecoder pipeline. Using these NCBI RefSeq representative protein-coding gene models as a reference, the StringTie assembled a total of 26,061 gene models with 41,390 transcripts. Among them, 14,005 gene models containing 29,053 novel transcripts (53.74% of total gene models) had no overlaps with the NCBI RefSeq representative protein-coding gene models, and were defined as candidate novel genes. The candidate novel gene sequences were scanned directly for CDSs with TransDecoder (Haas, 2014), which generated 1,939 novel protein-coding gene models after filtering out low-quality CDSs (see filtering criteria in the Methods). Together with a manually annotated cytochrome P450 gene (see next section), we identified 1,940 novel protein-coding gene models in the B. mandarina genome. When incorporating the NCBI RefSeq annotation, there were 14,212 protein-coding gene models for the B. mandarina genome.
Among these 1,940 novel protein-coding genes, 1,295 genes (66.75%) could be functionally annotated to at least one of the four databases, including NR (non-redundant protein sequences in NCBI), SwissProt (Boeckmann et al., 2003), KEGG, and GO. For the whole B. mandarina proteome, approximately 94.38% of genes could be functionally annotated compared with the 95.14% in B. mori.
The identification and classification of cytochrome P450 genes in B. mandarina
By integrating the results from homologous to B. mori cytochrome P450 proteins and the existence of complete cytochrome P450 domain (PF00067), we identified 67 cytochrome P450 genes in the B. mandarina genome. To find all of the cytochrome P450-related genes in the genomes, we further manually annotated the genome using the B. mori cytochrome P450 proteins as a reference (see Methods section). This homology-based gene annotation strategy detected one additional P450 gene, resulting in a total of 68 cytochrome P450 genes in the B. mandarina genome. For B. mori, Ai et al. (2011) identified 84 cytochrome P450 sequences based on a silkworm draft genome, while Kawamoto et al. (2019) identified 83 cytochrome P450 genes with a high-quality genome assembly and a unified set containing 84 cytochrome P450 genes was used in our downstream analyses (Table 1).
| B. mandarina | B. mori | Gene | AA length | Exon number | Identity (%) |
|---|---|---|---|---|---|
| CYP2Clan | |||||
| XP_028039023.1 | KWMTBOMO06658 | CYP15C1 | 390 | 8 | 98.71 |
| XP_028040961.1 | KWMTBOMO05795 | CYP18A1 | 541 | 7 | 100.00 |
| XP_028040963.1 | KWMTBOMO05796 | CYP18B1 | 533 | 7 | 99.15 |
| XP_028034668.1 | KWMTBOMO00033 | CYP303A1 | 498 | 3 | 99.00 |
| MSTRG.449.1 | KWMTBOMO00224 | CYP305B1 | 487 | 9 | 100.00 |
| XP_028040956.1 | KWMTBOMO05794 | CYP306A1 | 538 | 8 | 100.00 |
| XP_028041590.1 | KWMTBOMO06147 | CYP307A1 | 536 | 2 | 99.51 |
| CYP3Clan | |||||
| XP_028040226.1 | KWMTBOMO12795 | CYP324A1 | 499 | 9 | 99.60 |
| XP_028035792.1 | KWMTBOMO08858 | CYP332A1 | 500 | 8 | 99.58 |
| XP_028039903.1 | KWMTBOMO09327 | CYP337A2 | 488 | 2 | 98.98 |
| XP_028039890.1 | KWMTBOMO09329 | CYP337A1 | 489 | 2 | 98.57 |
| XP_028030266.1 | KWMTBOMO07090 | CYP338A1 | 464 | 2 | 98.28 |
| XP_028037108.1 | KWMTBOMO04531 | CYP354A1 | 516 | 9 | 99.03 |
| – | KWMTBOMO12722 | CYP365A1 | – | – | – |
| XP_028030488.1 | KWMTBOMO06852 | CYP6AB5 | 513 | 2 | 98.44 |
| MSTRG.11650.2 | KWMTBOMO12342 | CYP6AB4 | 521 | 2 | 91.77 |
| – | KWMTBOMO12343 | CYP6AB8 | – | – | – |
| XP_028037754.1 | KWMTBOMO07237 | CYP6AE9 | 517 | 2 | 97.87 |
| XP_028026771.1 | KWMTBOMO09944* | CYP6AE7 | 515 | 2 | 97.67 |
| XP_028026792.1 | KWMTBOMO09944* | CYP6AE7 | 523 | 2 | 85.24 |
| XP_028026744.1 | KWMTBOMO09945 | CYP6AE6P | 515 | 4 | 100.00 |
| – | KWMTBOMO09947 | CYP6AE5 | – | – | – |
| – | KWMTBOMO09950 | CYP6AE5 | – | – | – |
| XP_028026755.1 | KWMTBOMO09951-1M | CYP6AE4 | 523 | 2 | 94.22 |
| XP_028026857.1 | KWMTBOMO09951-2M | CYP6AE2 | 523 | 2 | 99.04 |
| XP_028026790.1 | KWMTBOMO09952$ | CYP6AE3P | 523 | 2 | 96.56 |
| XP_028026883.1 | KWMTBOMO09952$ | CYP6AE3P | 523 | 2 | 96.56 |
| XP_028033575.1 | KWMTBOMO13805 | CYP6AE22 | 516 | 2 | 99.81 |
| MSTRG.12374.1 | KWMTBOMO13412 | CYP6AN2 | 515 | 3 | 99.21 |
| XP_028037243.1 | KWMTBOMO12654 | CYP6AU1 | 496 | 2 | 99.80 |
| CYP6AV1-Bman | CYP6AV1 | CYP6AV1 | 500 | 2 | 99.800 |
| – | KWMTBOMO05640 | CYP6AW1 | – | – | – |
| XP_028027733.1 | KWMTBOMO12622 | CYP6B29 | 505 | 2 | 100.00 |
| XP_028036989.1 | KWMTBOMO10620 | CYP9A20 | 531 | 10 | 98.87 |
| XP_028036963.1 | KWMTBOMO10621-1M | CYP9A19 | 490 | 10 | 90.67 |
| XP_028042624.1 | KWMTBOMO10621-2M | CYP9A21 | 158 | 3 | 83.70 |
| XP_028036964.1 | KWMTBOMO10626 | CYP9A22 | 531 | 11 | 98.87 |
| XP_028033856.1 | KWMTBOMO10600 | CYP9AJ1 | 505 | 10 | 98.01 |
| XP_028035894.1 | KWMTBOMO09346 | CYP9G1 | 495 | 9 | 92.53 |
| XP_028036982.1 | KWMTBOMO10603 | CYP9G3 | 525 | 10 | 99.43 |
| CYP4Clan | |||||
| – | KWMTBOMO15697 | CYP340A1 | – | – | – |
| – | KWMTBOMO15698 | CYP340A5P | – | – | – |
| XP_028040572.1 | KWMTBOMO15699 | CYP340A4 | 488 | 10 | 97.10 |
| XP_028040567.1 | KWMTBOMO15700 | CYP340A2 | 460 | 9 | 95.38 |
| – | KWMTBOMO15704-15705M | CYP340A6 | – | – | – |
| XP_028040566.1 | KWMTBOMO15707 | CYP340A3 | 489 | 10 | 99.80 |
| XP_028043542.1 | KWMTBOMO15837 | CYP340B1 | 484 | 10 | 96.23 |
| XP_028043540.1 | KWMTBOMO15838 | CYP340C1 | 495 | 11 | 99.02 |
| XP_028040570.1 | KWMTBOMO15694 | CYP340D1 | 487 | 10 | 100.00 |
| – | KWMTBOMO15695 | CYP340E1 | – | – | – |
| XP_028031815.1 | KWMTBOMO15685 | CYP340F1 | 491 | 10 | 98.45 |
| XP_028043541.1 | KWMTBOMO15835 | CYP340un1 | 487 | 12 | 97.94 |
| – | CYP341A6 | CYP341A6 | – | – | – |
| XP_028036361.1 | KWMTBOMO13274-13275M | CYP341A1 | 508 | 10 | 98.23 |
| XP_028036360.1 | KWMTBOMO13276 | CYP341A3 | 504 | 10 | 94.82 |
| XP_028036359.1 | KWMTBOMO13278 | CYP341A4 | 508 | 10 | 85.32 |
| XP_028036358.1 | KWMTBOMO13279 | CYP341A5 | 508 | 10 | 99.16 |
| XP_028036416.1 | KWMTBOMO13280 | CYP341A7 | 379 | 8 | 98.33 |
| XP_028040208.1 | KWMTBOMO13324 | CYP341B1 | 512 | 10 | 99.34 |
| XP_028026371.1 | KWMTBOMO13451 | CYP341C1 | 505 | 10 | 99.51 |
| MSTRG.17906.1 | KWMTBOMO01080 | CYP366A1 | 557 | 11 | 99.55 |
| – | KWMTBOMO09792 | CYP367A1 | – | – | – |
| XP_028036546.1 | KWMTBOMO09791 | CYP367B1 | 497 | 10 | 96.58 |
| XP_028041506.1 | KWMTBOMO01330 | CYP4AU2 | 495 | 10 | 99.09 |
| – | KWMTBOMO12747 | CYP4AX1 | – | – | – |
| – | KWMTBOMO12748 | CYP4AX2 | – | – | – |
| XP_028042103.1 | KWMTBOMO07943 | CYP4G22 | 556 | 9 | 100.00 |
| XP_028033176.1 | KWMTBOMO07978 | CYP4G24 | 562 | 11 | 94.61 |
| XP_028033210.1 | KWMTBOMO07979-1M | CYP4G23 | 562 | 10 | 99.47 |
| – | KWMTBOMO07979-2M | CYP4G23 | – | – | – |
| XP_028028458.1 | KWMTBOMO07690 | CYP4L6 | 499 | 11 | 98.33 |
| – | KWMTBOMO02817 | CYP4M9 | – | – | – |
| XP_028040841.1 | KWMTBOMO02818 | CYP4M5 | 503 | 9 | 99.80 |
| XP_028042717.1 | KWMTBOMO12746 | CYP4S5 | 125 | 1 | 100.00 |
| – | KWMTBOMO12749 | CYP4S6 | – | – | – |
| Mito.Clan | |||||
| XP_028034514.1 | KWMTBOMO08259 | CYP301A1 | 528 | 8 | 100.00 |
| XP_028032374.1 | KWMTBOMO13168 | CYP302A1 | 517 | 8 | 98.45 |
| XP_028030999.1 | KWMTBOMO03959-03960M | CYP314A1 | 516 | 9 | 92.49 |
| XP_028033300.1 | KWMTBOMO04611 | CYP315A1 | 496 | 8 | 99.58 |
| XP_028042905.1 | KWMTBOMO04516 | CYP333A2 | 420 | 9 | 98.81 |
| XP_028028513.1 | KWMTBOMO07693 | CYP333B2 | 510 | 10 | 99.22 |
| XP_028028461.1 | KWMTBOMO07694 | CYP333B1 | 512 | 10 | 99.41 |
| – | KWMTBOMO04339 | CYP333un1 | – | – | – |
| XP_028043257.1 | KWMTBOMO11023-11024M | CYP339A1 | 577 | 10 | 99.67 |
| XP_028042777.1 | KWMTBOMO08262 | CYP49A1 | 424 | 10 | 99.37 |
| – | KWMTBOMO11585 | CYP49A2 | – | – | – |
Although B. mandarina has a significantly smaller cytochrome P450 repository than B. mori, we did find two duplicated genes in the B. mandarina. These genes are all belong to CYP6AE subfamily members. Interestingly, in the cotton bollworm Helicoverpa armigera, CYP6AE gene cluster knockout using the CRISPR-Cas9-based genome editing tools reveals their roles in detoxification of phytochemicals and insecticides (Wang et al., 2018). Therefore, the duplicated CYP6AE subfamily members in the B. mandarina may contribute to their reduced susceptibility to the insecticides used for control (Bing et al., 2010).
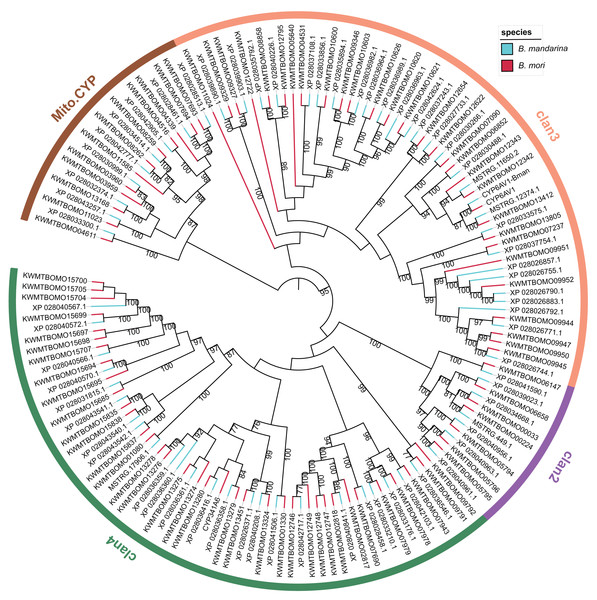
We constructed a maximum likelihood tree using the P450 proteins from B. mandarina and B. mori (Fig. 2). Consistent with previous results, these P450 genes can be grouped into four major clades, which are common in the insects and include the CYP2, CYP3, CYP4 and mitochondrial CYP clades. Using the phylogenetic tree, the 68 P450 genes in B. mandarina were classified into 25 families and 45 subfamilies according to the standard nomenclature and classification of P450 genes in B. mori. When compared with B. mori, the CYP365 family is missing in B. mandarina. For the two cytochrome P450 genes in B. mori identified by Ai et al. but missing in the Kawamoto et al. (Ai et al., 2011), the CYP6AV1 is present in B. mandarina, but the CYP341A6 is missing.
Figure 2: Phylogenetic tree of the cytochrome P450 genes in the wild and domestic silkworms.
The bootstrap value for 100 trials is labeled on each branch (only values ≥ 75 are shown). The families attributed to insect P450 genes are marked for each clan on the circle.The structural divergence of gene family members may arise due to exon/intron loss or gain and other mechanisms, and analyses of exon/intron structures can be important in revealing the evolutionary history of the gene family. The investigation of the intron–exon organizations of B. mandarina cytochrome P450 genes revealed highly variable intron-exon structures among these genes (Fig. S1). However, the genes in a clade of the CYP3 clan, which is the largest clan and is most closely related to vertebrate CYP3 and CYP5 families, have significantly smaller numbers of exons and a longer length for each exon than those in the other clans. These similarities in exon−intron organization in this clade provide a strong support for a common origin.
Genomic distribution of P450 superfamily in the silkworm
Based on the physical maps, we located 66 cytochrome P450s on 18 different chromosomes in B. mandarina. Only two genes were on the scaffolds. The XP_028042624.1 (CYP9A21) was on the scaffold NW_021012135.1, while its B. mori ortholog was on chromosome 17. The XP_028042717.1 (CYP4S5) was on scaffold NW_021013128.1, while its ortholog in the B. mori was on chromosome 21.
The genomic distribution patterns of the cytochrome P450 genes in these two silkworm species are different. For B. mandarina, 27 cytochrome P450 genes were concentrated into 7 clusters, which are defined as containing at least three genes, while for B. mori, 37 cytochrome P450 genes were concentrated into 8 clusters (Fig. 3). B. mori has one more cluster, which is located on chromosome 21 and has four genes. Three of the four genes were not found in B. mandarina, indicating potential loss. The largest single cluster in B. mori is located on chromosome 26, which has 9 CYP340 genes; however, the synteny regions in B. mandarina only have 4 CYP340 genes.
Figure 3: Physical locations of B. mandarina (A) and B. mori (B) P450 genes on the chromosomes.
Chromosomes are grey rectangles, bold number marked on each chromosome is their chromosome number, and ticks on each chromosome are the physical positions (unit: Mbp). P450 genes are colored according their clan group information.Figure 3 shows that these two species also share common characteristics in the genomic distribution of cytochrome P450 genes. The P450s are unevenly distributed in the genome. Most of the cytochrome P450s are tandemly arranged on chromosomes in both silkworm species. All of the CYP340 genes are located on chromosome 26 and form at least two clusters. All of the CYP341 genes are located on chromosome 22, and form a cluster and a singleton. CYP9 genes are located in the chromosome 17 and form a cluster, with the exception of CYP9G1, which is located on chromosome 15 as a singleton.
Computational functional analyses of B. mandarina cytochrome P450 genes
To explore the functional roles of the cytochrome P450 genes in B. mandarina, we obtained the GO terms from Gene Ontology (GO), which classifies genes into three GO categories: cellular component, molecular function, and biological process. Using the topGO (Alexa & Rahnenfuhrer, 2010) package in R programming language (https://www.r-project.org/), we identified four over-represented GO terms for molecular function and one over-represented term for biological process (Table S6). For the molecular function, there were 67 of 107 cytochrome P450 genes annotated to the GO:0020037 (heme binding, adjusted p-value: 0) and 3 of 16 cytochrome P450 genes annotated to the GO:0004497 (monooxygenase activity, adjusted p-value = 0.045; Table S6). These results are consistent with the fact that the cytochrome P450 genes are heme-containing monooxygenases.
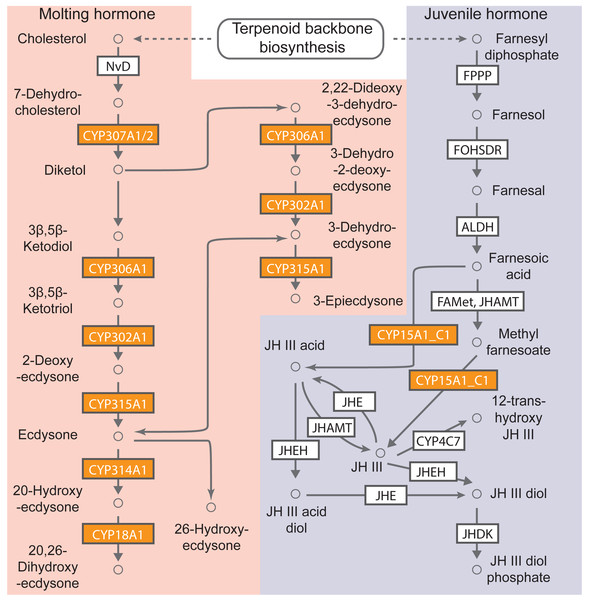
KEGG pathway-based analysis was also performed to determine the biological functions of cytochrome P450 genes in B. mandarina. A total of 63 cytochrome P450 genes (92.65% of total cytochrome P450 genes) could be assigned to 13 non-redundant KO numbers using the KOBAS (Table S7). Among these KO orthologs, the five most abundant KOs were K14999 (gene number: 20, cytochrome P450 family 6 [EC:1.14.-.-]) and K15001 (gene number: 19, cytochrome P450 family 4 [EC:1.14.-.-]). Using KEGG mapper, 7 of these 13 KOs were mapped to the map00981 (Insect hormone biosynthesis), and in our KEGG enrichment analysis, this pathway was also significantly enriched with an adjusted p value of 4.48e−18 (Fig. 4). These results revealed the involvement of cytochrome P450 genes in insect hormone biosynthesis.
Figure 4: B. mandarina cytochrome P450 genes were significantly enriched in the insect hormone biosynthesis pathway.
This diagram of insect hormone biosynthesis pathway is adapted from KEGG (map00981), and the boxes colored with yolk yellow are the cytochrome P450 genes found in B. mandarina. FPPP, farnesyl diphosphate phosphatase; FOHSDR, NADP+-dependent farnesol dehydrogenase; ALDH, aldehyde dehydrogenase (NAD+); FAMeT, farsoic acid methyltransferase; JHAMT, juvenile hormone-III synthase; JHE, juvenile-hormone esterase; JHEH, juvenile hormone epoxide hydrolase; JHDK, juvenile hormone diol kinase; NvD, cholesterol 7-desaturase.Expression pattern of cytochrome P450 genes in silkworms
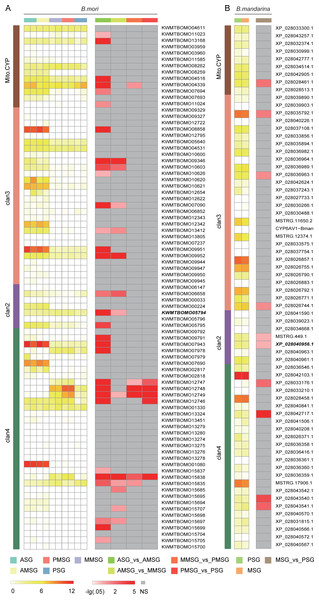
To explore cytochrome P450 gene expression in the silk gland, we collected two RNASeq datasets from silk gland, including the anterior silk gland (ASG), anterior median silk gland (AMSG), middle MSG (MMSG), posterior MSG (PMSG) and posterior silk gland (PSG) (Chang et al., 2015; Cheng et al., 2015). A total of 43 (63% of 68) and 76 (90% of 84) cytochrome P450 genes were expressed (raw counts ≥ 2) in at least one tissue of B. mandarina and B. mori, respectively. Totally, we identified 10 differentially expressed genes (DEGs) and 32 DEGs in B. mandarina and B. mori tissues respectively (Fig. 5).
Figure 5: Expression analysis of cytochrome P450 genes across different tissues in B. mori (A) and B. mandarina (B).
The first column is the log10-transformed TMM (trimmed mean of M-values)-normalized expression data, and the tissue types are marked at the top of the heatmap. The second column is the –log10-transformed false discovery rate (FDR) value of DEG analyses from edgeR, and the comparaison scheme are also marked at the top of the heatmap. NS: not significant, namely, FDR > 0.05. Gene ID in Bold style is the CYP306A1.Discussion
By implementing a reference-assisted approach, we built chromosomes from genome contigs or scaffolds published by Xiang et al. (2018) for B. mandarina. The results revealed high genome synteny but also abundant structural rearrangements among the silkworms (Fig. 1). Despite building from a straight-forward alignment-based approach, our reference-assisted chromosome level assemblies can be used in downstream comparative genomic analysis and other types of analyses. We also updated the gene annotation using StringTie-TransDecoder pipeline, and this pipeline identified 1,940 novel protein-coding gene models in the B. mandarina genome, indicating the necessity of updating the gene annotation.
We identified 67 cytochrome P450 genes in the B. mandarina genome (Table 1). The genomic distribution patterns of the cytochrome P450 genes in these two silkworm species are different (Fig. 3). Computational functional analyses of B. mandarina cytochrome P450 genes revealed the involvement of cytochrome P450 genes in insect hormone biosynthesis (Fig. 4). The P450 repository in B. mandarina is smaller than in B. mori and other insects, except for Vanessa tameamea (Table S5 ). This suggests loss of cytochrome P450 genes during evolution. Although B. mandarina is very similar to B. mori in physiological and morphological characteristics, resistance to insecticides differs between the two species due to natural selection. B. mori are weakly resistant to insecticides, and silk production is reduced by >30% annually in China because of insecticide poisoning. In contrast, B. mandarina is a major pest of mulberry and are showing reduced susceptibility to the insecticides used for control (Bing et al., 2010). Cytochrome P450 genes play a major role in insecticide resistance, allowing faster metabolic removal of insecticides (Feyereisen, 2015). However, the number of P450 genes in B. mandarina is much lower than that in B. mori, suggesting that selected key genes, rather than the total gene number of cytochrome P450s, are related to the increased resistance to insecticide resistance in B. mandarina.
Silk production is very different in B. mandarina and B. mori (Chang et al., 2015), and this phenotype attracted many attentions and accumulated many sequencing datasets (Chang et al., 2015; Cheng et al., 2015). The cytochrome P450 enzymes are found in almost all insect tissues. They fulfill many important tasks, from the synthesis and degradation of ecdysteroids and JHs to insecticide metabolism (Feyereisen, 1999). Therefore, it is important to study the expression of cytochrome P450 genes in the silk glands of B. mandarina and B. mori. The silk gland is the only organ that produces silk proteins (fibroins and sericins). The silk gland is divided into three main parts: anterior silk gland (ASG), median silk gland (MSG), and posterior silk gland (PSG). The MSG can be further divided into anterior, middle, and posterior MSG (AMSG, MMSG, and PMSG, respectively) (Chang et al., 2015). PSGs are responsible for the synthesis and secretion of fibroins, MSG synthesize the sericins (glue proteins), and the ASG processes the liquid silk proteins and secretes them during cocoon formation (Fang et al., 2015). Two RNASeq datasets from the above tissues (Chang et al., 2015; Cheng et al., 2015) were used to explore cytochrome P450 gene expression in the silk gland.
The silk gland is greatly affected by insect hormones, especially, ecdysone and juvenile hormone (JH). Growth and differentiations of the silk gland cells are accelerated by ecdysone, and are controlled by JH (Akai, 1979). Daimon et al. revealed the essential role of CYP15C1 for the JH biosynthesis, and found that this gene is specifically expressed in the corpus allatum, an endocrine organ that synthesizes and secretes JHs (Daimon et al., 2012). However, we found that this gene is also expressed in the silk gland, although at a very low expression level (Fig. 5). Cheng Dao-Jun et al. (2014) found four cytochrome P450 genes involved in ecdysteroidogenesis, including CYP306A1, CYP302A1, CYP315A1, and CYP314A1. Among these four genes, the CYP306A1 was found significantly differently expressed in both B. mandarina (XP_028040956.1) and B. mori (KWMTBOMO05794). CYP302A1 was only found to be significantly differently expressed in B. mori (KWMTBOMO13168) but not in B. mandarina (XP_028032374.1), while the CYP315A1 (KWMTBOMO04611 for B. mori, XP_028033300.1 for B. mandarina), and CYP314A1 (KWMTBOMO03959-03960M for B. mori, XP_028030999.1 for B. mandarina) were not differently expressed in the two silkworms (Fig. 5).
Phoxim exposure is toxic to silkworms, causes a decrease of fibroin synthesis, and affects silk production. After phoxim exposure, Cheng et al. (2018) found that the transcriptional levels of CYP6AB and CYP306A were up-regulated by 1.731- and 1.221-fold, respectively. There are two and three CYP6AB genes in B. mandarina (XP_028030488.1, MSTRG.11650.2) and B. mori (KWMTBOMO06852, KWMTBOMO12342, and KWMTBOMO12343), respectively; however, they are not significantly expressed. Interestingly, CYP306A was found to be significantly differently expressed in both B. mandarina (XP_028040956.1) and B. mori (KWMTBOMO05794) (Fig. 5). We also checked the expression of CYP306A in the midgut from the B. mandarina, which tissues is one of the major tissues for insecticides metabolization. We blasted the CYP306A sequences to the RNASeq assemblies in the SilkBase (Kawamoto, 2017), and the expression was validated by its homologous sequences in the SilkBase (A_BomaMG_comp25068_c0_seq1 and A_BomaMG_comp25068_c0_seq2).
Conclusions
In this study, we improved the quality of the genome assemblies and updated the protein-coding gene annotations for B. mandarina using the genome of B. mori as reference. Using in silico analyses of B. mandarina genomes, we identified 68 cytochrome P450 genes. Comparison with other insects revealed that B. mandarina may have lost many cytochrome P450 genes. Analyses of the silk gland transcriptome identified candidate cytochrome P450 genes (such as CYP306A) involved in ecdysteroidogenesis and insecticide metabolism in B. mandarina. Altogether, these results provided a genome-wide glimpse of the B. mandarina cytochrome P450 repository; however, the up- or down-regulated cytochrome P450 genes require more wet experiments to explore their biological roles.