Subcellular Localization of the TFF Peptides xP1 and xP4 in the Xenopus laevis Gastric/Esophageal Mucosa: Different Secretion Modes Reflecting Diverse Protective Functions
Abstract
:1. Introduction
2. Results
2.1. Localization of xP1 in the X. laevis Gastric Mucosa
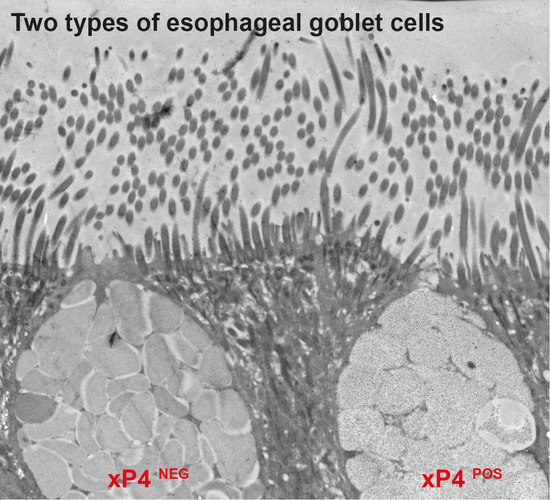
2.2. Localization of xP4 in the X. laevis Esophagus
3. Discussion
3.1. Secretion of xP1 in Gastric Surface Mucous Cells: Concentration in Dense Core Regions of Secretory Granules
3.2. Secretion of xP4 in Esophageal Goblet Cells: Different Types of Secretory Granules and Goblet Cells
4. Materials and Methods
4.1. Preparation of Tissue Samples for Immunofluorescence and Immunoelectron Microscopy
4.2. Lectins and Antisera
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| DAPI | 4’,6-diamidino-2-phenylindole dihydrochloride |
| TFF | Trefoil factor family |
References
- Hauser, F.; Hoffmann, W. xP1 and xP4. P-domain peptides expressed in Xenopus laevis stomach mucosa. J. Biol. Chem. 1991, 266, 21306–21309. [Google Scholar] [PubMed]
- Hoffmann, W.; Hauser, F. The P-domain or trefoil motif: a role in renewal and pathology of mucous epithelia? Trends Biochem. Sci. 1993, 18, 239–243. [Google Scholar] [CrossRef]
- Hoffmann, W.; Jagla, W. Cell type specific expression of secretory TFF peptides: colocalization with mucins and synthesis in the brain. Int. Rev. Cytol. 2002, 213, 147–181. [Google Scholar] [PubMed]
- Hoffmann, W. TFF Peptides. In Handbook of Biologically Active Peptides, 2nd ed.; Kastin, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Hoffmann, W. Trefoil factor family (TFF) peptides: regulators of mucosal regeneration and repair, and more. Peptides 2004, 727–730. [Google Scholar] [CrossRef]
- Braga Emidio, N.; Hoffmann, W.; Brierly, S.M.; Muttenthaler, M. Trefoil factor family: unresolved questions and clinical perspectives. Trends Biochem. Sci. 2019, 44, 387–390. [Google Scholar] [CrossRef]
- Jagla, W.; Wiede, A.; Kolle, S.; Hoffmann, W. Differential expression of the TFF-peptides xP1 and xP4 in the gastrointestinal tract of Xenopus laevis. Cell Tissue Res. 1998, 291, 13–18. [Google Scholar] [CrossRef]
- Ikuzawa, M.; Yasumasu, S.; Kobayashi, K.; Iuchi, I. Cloning and expression of xP1-L, a new marker gene for larval surface mucous cells of tadpole stomach in Xenopus laevis. Gene Expr. Patterns 2007, 8, 12–18. [Google Scholar] [CrossRef]
- Botzler, C.; Oertel, M.; Hinz, M.; Hoffmann, W. Structure of the Xenopus laevis TFF-gene xP4.1, differentially expressed to its duplicated homolog xP4.2. Biochim. Biophys. Acta 1999, 1489, 345–353. [Google Scholar] [CrossRef]
- Session, A.M.; Uno, Y.; Kwon, T.; Chapman, J.A.; Toyoda, A.; Takahashi, S.; Fukui, A.; Hikosaka, A.; Suzuki, A.; Kondo, M.; et al. Genome evolution in the allotetraploid frog Xenopus laevis. Nature 2016, 538, 336–343. [Google Scholar] [CrossRef] [Green Version]
- Stürmer, R.; Reising, J.; Hoffmann, W. The TFF Peptides xP1 and xP4 Appear in Distinctive Forms in the Xenopus laevis Gastric Mucosa: Indications for Different Protective Functions. Int. J. Mol. Sci. 2019, 20, 6052. [Google Scholar] [CrossRef] [Green Version]
- Lefebvre, O.; Chenard, M.P.; Masson, R.; Linares, J.; Dierich, A.; LeMeur, M.; Wendling, C.; Tomasetto, C.; Chambon, P.; Rio, M.C. Gastric mucosa abnormalities and tumorigenesis in mice lacking the pS2 trefoil protein. Science 1996, 274, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Tomasetto, C.; Rio, M.C. Pleiotropic effects of Trefoil Factor 1 deficiency. Cell Mol. Life Sci. 2005, 62, 2916–2920. [Google Scholar] [CrossRef] [PubMed]
- Stürmer, R.; Harder, S.; Schlüter, H.; Hoffmann, W. Commercial Porcine Gastric Mucin Preparations, also Used as Artificial Saliva, are a Rich Source for the Lectin TFF2: In Vitro Binding Studies. ChemBioChem. 2018, 19, 2598–2608. [Google Scholar] [CrossRef] [PubMed]
- Heuer, F.; Stürmer, R.; Heuer, J.; Kalinski, T.; Lemke, A.; Meyer, F.; Hoffmann, W. Different forms of TFF2, a lectin oft he human gastric mucus barrier: in vitro binding studies. Int. J. Mol. Sci. 2019, 20, 5871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoffmann, W. TFF2, a MUC6-binding lectin stabilizing the gastric mucus barrier and more. Int. J. Oncool. 2015, 47, 806–816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ishihara, K.; Kurihara, M.; Goso, Y.; Urata, T.; Ota, H.; Katsuyama, T.; Hotta, K. Peripheral α-linked N-acetylglucosamine on the carbohydrate moiety of mucin derived from mammalian gastric gland mucous cells: epitope recognized by a newly characterized monoclonal antibody. Biochem. J. 1996, 318, 409–416. [Google Scholar] [CrossRef]
- Oinuma, T.; Kawano, J.-I.; Suganama, T. Glycoconjugate histochemistry of Xenopus laevis fundic gland with special reference to mucous neck cells during development. Anat. Rec. 1991, 230, 502–512. [Google Scholar] [CrossRef]
- Nakayama, J. Dual roles of gastric gland mucin-specific O-glycans in prevention of gastric cancer. Acta Histochem. Cytochem. 2014, 47, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Lang, T.; Klasson, S.; Larsson, E.; Johansson, M.E.V.; Hansson, G.C.; Samuelsson, T. Searching the evolutionary origin of epithelial mucus protein components – mucins and FCGBP. Mol. Biol. Evol. 2016, 33, 1921–1936. [Google Scholar] [CrossRef]
- Fox, J.G.; Rogers, A.B.; Whary, M.T.; Ge, Z.; Ohtani, M.; Jones, E.K.; Wang, T.C. Accelerated progression of gastritis to dysplasia in the pyloric antrum of TFF2 -/- C57BL6 x Sv129 Helicobacter pylori-infected mice. Am. J. Pathol. 2007, 171, 1520–1528. [Google Scholar] [CrossRef] [Green Version]
- Rio, M.C.; Chenard, M.P.; Wolf, C.; Marcellin, L.; Tomasetto, C.; Lathe, R.; Bellocq, J.P.; Chambon, P. Induction of pS2 and hSP genes as markers of mucosal ulceration of the digestive tract. Gastroenterology 1991, 100, 375–379. [Google Scholar] [CrossRef]
- Lefebvre, O.; Wolf, C.; Kédinger, M.; Chenard, M.-P.; Tomasetto, C.; Chambon, P.; Rio, M.-C. The mouse one P-domain (pS2) and two P-domain (mSP) genes exhibit distinct patterns of expression. J. Cell Biol. 1993, 122, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Hanby, A.M.; Poulsom, R.; Singh, S.; Elia, G.; Jeffery, R.E.; Wright, N.A. Spasmolytic polypeptide is a major antral peptide: Distribution of the trefoil peptides human spasmolytic polypeptide and pS2 in the stomach. Gastroenterology 1993, 105, 1110–1116. [Google Scholar] [CrossRef]
- Jeffrey, G.P.; Oates, P.S.; Wang, T.C.; Babyatsky, M.W.; Brand, S.J. Spasmolytic polypeptide: A trefoil peptide secreted by rat gastric mucous cells. Gastroenterology 1994, 106, 336–345. [Google Scholar] [CrossRef]
- Byrd, J.C.; Yan, P.; Sternberg, L.; Yunker, C.K.; Scheiman, J.M.; Bresalier, R.S. Aberrant expression of gland-type gastric mucin in the surface epithelium of Helicobacter pylori-infected patients. Gastroenterology 1997, 113, 455–464. [Google Scholar] [CrossRef]
- Karam, S.M.; Tomasetto, C.; Rio, M.C. Trefoil factor 1 is required for the commitment programme of mouse oxyntic epithelial progenitors. Gut 2004, 53, 1408–1415. [Google Scholar] [CrossRef] [Green Version]
- Schumacher, U.; Adam, E.; Hauser, F.; Probst, J.C.; Hoffmann, W. Molecular anatomy of a skin gland: Histochemical and biochemical investigations on the mucous glands of Xenopus laevis. J. Histochem. Cytochem. 1994, 42, 57–65. [Google Scholar] [CrossRef]
- Perez-Vilar, J. Mucin granule intraluminal organization. Am. J. Respir. Cell Mol. Biol. 2007, 36, 183–190. [Google Scholar] [CrossRef] [Green Version]
- Perez-Vilar, J.; Olsen, J.C.; Chua, M.; Boucher, R.C. pH-dependent intraluminal organization of mucin granules in live human mucous/goblet cells. J. Biol. Chem. 2005, 280, 16868–16881. [Google Scholar] [CrossRef] [Green Version]
- Perez-Vilar, J.; Mabolo, R.; McVaugh, C.T.; Bertozzi, C.R.; Boucher, R.C. Mucin granule intraluminal organization in living mucous/goblet cells. Roles of protein post-translational modifications and secretion. J. Biol. Chem. 2006, 281, 4844–4855. [Google Scholar] [CrossRef] [Green Version]
- Thim, L.; Madsen, F.; Poulsen, S.S. Effect of trefoil factors on the viscoelastic properties of mucus gels. Eur. J. Clin. Invest. 2002, 32, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Carlemalm, E.; Villiger, W.; Hobot, J.A.; Acetarin, J.D.; Kellenberger, E. Low temperature embedding with Lowicryl resins: two new formulations and some applications. J. Microsc. 1985, 140, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, H.; Humbel, B.M. Correlative light and electron microscopy using immunolabeled resin sections. Methods Mol. Biol. 2007, 369, 229–256. [Google Scholar] [PubMed]
- Schwarz, H.; Humbel, B.M. Correlative light and electron microscopy using immunolabeled sections. Methods Mol. Biol. 2014, 1117, 559–592. [Google Scholar] [PubMed]


| Goblet Cell | Granules | GSA-II/xP4 |
|---|---|---|
| Uniform-type (xP4-positive) | Type 1 | GSA-IIPOS/xP4POS |
| Halo-type (xP4-negative) | Type 2 Type 3 | GSA-IIPOS/xP4NEG GSA-IINEG/xP4NEG |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schwarz, H.; Hoffmann, W. Subcellular Localization of the TFF Peptides xP1 and xP4 in the Xenopus laevis Gastric/Esophageal Mucosa: Different Secretion Modes Reflecting Diverse Protective Functions. Int. J. Mol. Sci. 2020, 21, 761. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21030761
Schwarz H, Hoffmann W. Subcellular Localization of the TFF Peptides xP1 and xP4 in the Xenopus laevis Gastric/Esophageal Mucosa: Different Secretion Modes Reflecting Diverse Protective Functions. International Journal of Molecular Sciences. 2020; 21(3):761. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21030761
Chicago/Turabian StyleSchwarz, Heinz, and Werner Hoffmann. 2020. "Subcellular Localization of the TFF Peptides xP1 and xP4 in the Xenopus laevis Gastric/Esophageal Mucosa: Different Secretion Modes Reflecting Diverse Protective Functions" International Journal of Molecular Sciences 21, no. 3: 761. https://0-doi-org.brum.beds.ac.uk/10.3390/ijms21030761