Cellulose-Based Hydrogels as Sustained Drug-Delivery Systems
Abstract
:1. Introduction
- -
- the improvement of the targeted delivery of drugs to the specific site;
- -
- correlation of the drug release process with the patient’s circadian rhythm, a cycle of approximately 24 h that takes place at the biochemical, physiological and behavioral level, in response of the body to the light-dark alternation;
- -
- optimization of the water-soluble drugs release (reducing the release time);
- -
- increasing the bioavailability of medicines with a low solubility rate in water;
- -
- a carefully delivery control of the highly toxic drugs;
- -
- the use of more drugs in the same system;
- -
- improving the elimination of toxic compounds obtained from drug metabolism (fast elimination).
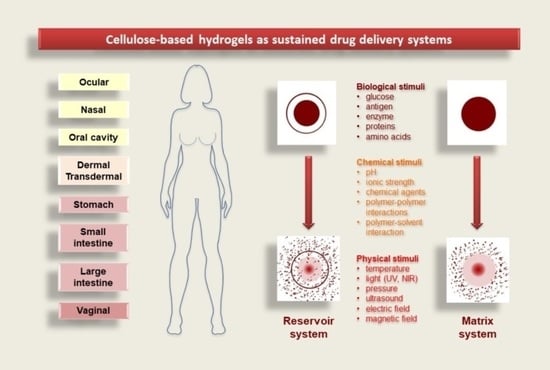
2. Controlled Drug-Delivery Locations
2.1. Dermal and Transdermal Drug Delivery
- -
- dermal administration—requires the crossing of the drug through the outer layer of the skin (stratum corneum), providing its location in the underlying layers of the skin;
- -
- transdermal delivery—the drug is transported to the skin dermis, followed by its access to the systemic circulation.
2.2. Ocular Drug Delivery
2.3. Nasal Drug Delivery
2.4. Gastrointestinal Tract Drug Delivery
2.5. Vaginal and Rectal Drug Delivery
2.6. Cancer Drug Delivery
3. Hydrogels as Potential Drug Delivery Systems
3.1. Properties of Hydrogels
- -
- macroporous—average pore size: 0.1–1 μm—drug release: mechanism that depends on the matrix porosity and diffusion coefficient of the drug;
- -
- microporous—average pore size: 100–1000 Å—drug release: molecular diffusion and convection;
- -
- non-porous—average pore size: 10–100 Å—drug release: diffusion mechanism.
3.2. Hydrogels Delivery Systems
3.2.1. Reservoir System
3.2.2. Matrix System
3.3. Hydrogel Drug-Release Mechanisms
- −
- diffusion-controlled—matrix and reservoir systems;
- −
- swelling-controlled—systems that are osmotically controlled and are activated with different solvents;
- −
- chemically controlled—systems that can be biodegraded or eroded and pendant chain systems.
3.3.1. Diffusion-Controlled Drug Mechanism
- -
- Rmesh/Rdrug > 1, mesh size is larger than the drug molecules: the whole release process is controlled by diffusion. It is the case of small drug molecules which diffuse freely through the network, and their migration is not dependent on the mesh size;
- -
- Rmesh/Rdrug ~ 1, mesh size reaches the drug size: the steric hindrance dominates the drug diffusion. The resulting effect is a slow drug diffusion, which is reflected by a slow and extended-release;
- -
- Rmesh/Rdrug < 1, mesh size is very small and/or drug molecules are too large. The effect of steric hindrance causes a blockage of the drug within the network, until there is a degradation of the network or an increase in mesh size by swelling or deformation.
3.3.2. Swelling-Controlled Drug-Release Mechanism
3.3.3. Chemically-Controlled Drug-Release Mechanism
4. Cellulose-Based Hydrogels in Drug Delivery
4.1. Cellulose and Cellulose Derivative-Based Hydrogels Properties
4.2. Cellulose-Based Hydrogels for Oral Administration
4.2.1. Oral Cavity Delivery
4.2.2. Stomach-Specific Drug Delivery
4.2.3. Colon-Specific Drug Delivery
4.3. Cellulose-Based Hydrogels for Dermal and Transdermal Administration
4.3.1. Dermal Administration
4.3.2. Transdermal Administration
4.4. Cellulose-Based Hydrogels for Ocular Administration
4.5. Cellulose-Based Hydrogels in Nasal Delivery
4.6. Cellulose-Based Hydrogels in Vaginal Delivery
4.7. Cellulose Based-Hydrogels as Delivery Systems in Cancer Therapy
4.8. Injectable Cellulose-Based Hydrogels in Drug Delivery
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gupta, A.K. Environmental responsive hydrogels: A novel approach in drug delivery system. J. Drug Deliv. Ther. 2012, 2, 1–8. [Google Scholar] [CrossRef]
- Moghanjoughi, A.A.; Khoshnevis, D.; Zarrabi, A. A concise review on smart polymers for controlled drug release. Drug Deliv. Transl. Res. 2016, 6, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.M.; Magalhães, M.; Veiga, F.; Figueiras, A. Cellulose-based hydrogels in topical drug delivery: A challenge in medical devices. In Cellulose-Based Superabsorbent Hydrogels, 1st ed.; Mondal, M.I.H., Ed.; Springer International Publishing AG: Basel, Switzerland, 2018; Chapter 40; pp. 1–29. [Google Scholar]
- Vashist, A.; Gupta, Y.; Ahmad, S. Interpenetrating biopolymer network based hydrogels for an effective drug delivery system. Carbohydr. Polym. 2012, 87, 1433–1439. [Google Scholar] [CrossRef]
- Kulkarni, R.V.; Biswanath, S. Electrically responsive smart hydrogels in drug delivery: A review. J. Appl. Biomater. Biomech. 2010, 5, 125–139. [Google Scholar]
- Gao, S.; Tang, G.; Hua, D.; Xiong, R.; Han, J.; Jiang, S.; Zhang, Q.; Huang, C. Stimuli-responsive bio-based polymeric systems and their applications. J. Mater. Chem. B 2019, 7, 709–729. [Google Scholar] [CrossRef]
- Ali, A.E.; El-Rehim, H.A.A.; Kamal, H.; Hegazy, D.E.A. Synthesis of Carboxymethyl Cellulose Based Drug Carrier Hydrogel Using Ionizing Radiation for Possible Use as Site Specific Delivery System. J. Macromol. Sci. Part A 2008, 45, 628–634. [Google Scholar] [CrossRef]
- Vlaia, L.; Coneac, G.; Olariu, I.; Lupuleasa, D. Cellulose-Derivatives-Based Hydrogels as Vehicles for Dermal and Transdermal Drug Delivery. In Emerging Concepts in Analysis and Applications of Hydrogels, 1st ed.; Majee, S.B., Ed.; IntechOpen: London, UK, 2016; Chapter 7; pp. 159–200. [Google Scholar]
- Simões, S.; Figueiras, A.; Veiga, F. Modular Hydrogels for Drug Delivery. J. Biomater. Nanobiotechnol. 2012, 3, 185–199. [Google Scholar] [CrossRef]
- Sood, N.; Bhardwaj, A.; Mehta, S.; Mehta, A. Stimuli-responsive hydrogels in drug delivery and tissue engineering. Drug Deliv. 2014, 23, 748–770. [Google Scholar] [CrossRef] [Green Version]
- Zhidong, L.; Li, J.; Nie, S.; Liu, H.; Ding, P.; Pan, W. Study of an alginate/HPMC-based in situ gelling ophthalmic delivery system for gatifloxacin. Int. J. Pharm. 2006, 315, 12–17. [Google Scholar] [CrossRef]
- Kumar, A.R.; Aeila, A.S.S. Sustained release matrix type drug delivery system: An overview. World J. Pharm. Pharm. Sci. 2019, 9, 470–480. [Google Scholar]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Hydrogels as drug delivery systems; Pros and cons. Trends Pharm. Sci. 2019, 5, 7–24. [Google Scholar]
- Kapoor, D.; Patel, M.; Vyas, R.B.; Lad, C.; Lal, B. Site Specific drug delivery through nasal route using bioadhesive polymers. J. Drug Deliv. Ther. 2015, 5, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Nokhodchi, A.; Raja, S.; Patel, P.; Asare-Addo, K. The Role of Oral Controlled Release Matrix Tablets in Drug Delivery Systems. BioImpacts 2012, 2, 175–187. [Google Scholar] [PubMed] [Green Version]
- Kumar, R.; Patil, M.B.; Patil, S.R.; Paschapur, M.S. Polysaccharides based colon specific drug delivery: A review. Int. J. Pharm. Tech. Res. 2009, 1, 334–346. [Google Scholar]
- Bai, J.P.; Burckart, G.J.; Mulberg, A.E. Literature Review of Gastrointestinal Physiology in the Elderly, in Pediatric Patients, and in Patients with Gastrointestinal Diseases. J. Pharm. Sci. 2016, 105, 476–483. [Google Scholar] [CrossRef] [Green Version]
- Barba, A.A.; D’Amore, M.; Chirico, S.; Lamberti, G.; Titomanlio, G. Swelling of cellulose derivative (HPMC) matrix systems for drug delivery. Carbohydr. Polym. 2009, 78, 469–474. [Google Scholar] [CrossRef]
- Edgar, K.J. Cellulose esters in drug delivery. Cellulose 2006, 14, 49–64. [Google Scholar] [CrossRef]
- Gharti, K.; Thapa, P.; Budhathoki, U.; Bhargava, A. Formulation and in vitro evaluation of floating tablets of hydroxypropyl methylcellulose and polyethylene oxide using ranitidine hydrochloride as a model drug. J. Young Pharm. 2012, 4, 201–208. [Google Scholar] [CrossRef] [Green Version]
- Dar, M.J.; Ali, H.; Khan, A.; Khan, G.M. Polymer-based drug delivery: The quest for local targeting of inflamed intestinal mucosa. J. Drug Target 2017, 25, 582–596. [Google Scholar] [CrossRef]
- Sudhakar, Y.; Kuotsu, K.; Bandyopadhyay, A. Buccal bioadhesive drug delivery—A promising option for orally less efficient drugs. J. Control. Release 2006, 114, 15–40. [Google Scholar] [CrossRef]
- Li, N.; Yu, M.; Deng, L.; Yang, J.; Wang, W. Thermosensitive hydrogel of hydrophobically-modified methylcellulose for intravaginal drug delivery. J. Mater. Sci. Mater. Med. 2012, 23, 1913–1919. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, N.B.; Goyal, A. Thermoreversible mucoadhesive insitu gel: A review. Int. J. Innov. Drug Discov. 2013, 3, 67–84. [Google Scholar]
- Cheng, W.; Gu, L.; Ren, W.; Liu, Y. Stimuli-responsive polymers for anti-cancer drug delivery. Mater. Sci. Eng. C 2014, 45, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Vicario-De-La-Torre, M.; Forcada, J. The Potential of Stimuli-Responsive Nanogels in Drug and Active Molecule Delivery for Targeted Therapy. Gels 2017, 3, 16. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Qi, X.; Chen, Y.; Wu, Z. Thermo-sensitive hydrogels for delivering biotherapeutic molecules: A review. Saudi Pharm. J. 2019, 27, 990–999. [Google Scholar] [CrossRef]
- Bawa, P.; Pillay, V.; Choonara, Y.E.; Du Toit, L.C. Stimuli-responsive polymers and their applications in drug delivery. Biomed. Mater. 2009, 4, 022001. [Google Scholar] [CrossRef]
- Onofrei, M.-D.; Filimon, A. Cellulose-based hydrogels: Designing concepts, properties, and perspectives for biomedical and environmental applications. In Polymer Science: Research Advances, Practical Applications and Educational Aspects; Mendez-Vilas, A., Solano, A., Eds.; Formatex Research Center S.L.: Badajoz, Spain, 2016; pp. 108–120. [Google Scholar]
- Zou, Y.; Zhang, L.; Yang, L.; Zhu, F.; Ding, M.; Lin, F.; Wang, Z.; Li, Y. “Click” chemistry in polymeric scaffolds: Bioactive materials for tissue engineering. J. Control. Release 2018, 273, 160–179. [Google Scholar] [CrossRef]
- Hu, J.; Yang, L.; Yang, P.; Jiang, S.; Liu, X.; Li, Y. Polydopamine free radical scavengers. Biomater. Sci. 2020, 8, 4940–4950. [Google Scholar] [CrossRef]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef] [Green Version]
- Nazar, H.; Roldo, M.; Fatouros, D.G.; Van Der Merwe, S.M.; Tsibouklis, J. Hydrogels in mucosal delivery. Ther. Deliv. 2012, 3, 535–555. [Google Scholar] [CrossRef]
- Amin, S.; Rajabnezhad, S.; Kohli, K. Hydrogels as potential drug delivery systems. Sci. Res. Essays 2009, 3, 1175–1183. [Google Scholar]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in Biology and Medicine: From Molecular Principles to Bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Lanzalaco, S.; Armelin, E. Poly(N-isopropylacrylamide) and Copolymers: A Review on Recent Progresses in Biomedical Applications. Gels 2017, 3, 36. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, C.; Pereira, P.A.C.; Gama, M. Self-Assembled Hydrogel Nanoparticles for Drug Delivery Applications. Materials 2010, 3, 1420–1460. [Google Scholar] [CrossRef] [Green Version]
- Sannino, A.; Demitri, C.; Madaghiele, M. Biodegradable Cellulose-based Hydrogels: Design and Applications. Materials 2009, 2, 353–373. [Google Scholar] [CrossRef]
- Cirillo, G.; Curcio, M.; Nicoletta, F.P.; Iemma, F. Injectable Hydrogels for Cancer Therapy over the Last Decade. Pharmaceutics 2019, 11, 486. [Google Scholar] [CrossRef] [Green Version]
- Pal, K.; Banthia, A.K.; Majumdar, D.K. Polymeric Hydrogels: Characterization and Biomedical Applications. Des. Monomers Polym. 2009, 12, 197–220. [Google Scholar] [CrossRef] [Green Version]
- Bruschi, M.L. Classification of therapeutic systems for drug delivery. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Bruschi, M.L., Ed.; Elsevier: Cambridge, UK, 2015; Chapter 3; pp. 29–36. [Google Scholar]
- Caccavo, D.; Cascone, S.; Lamberti, G.; Barba, A.A.; Larsson, A. Swellable hydrogel-based systems for controlled drug delivery. In Smart Drug Delivery System; Sezer, A.D., Ed.; IntechOpen Limited: London, UK, 2015; Chapter 10; pp. 2454–2712. [Google Scholar]
- Yang, W.-W.; Pierstorff, E. Reservoir-Based Polymer Drug Delivery Systems. J. Lab. Autom. 2012, 17, 50–58. [Google Scholar] [CrossRef]
- Sharma, K.; Singh, V.; Arora, A. Natural biodegradable polymers as matrices in transdermal drug delivery. Int. J. Drug Dev. Res. 2011, 3, 85–103. [Google Scholar]
- Patel, H.; Panchal, D.R.; Patel, U.; Brahmbhatt, T.; Suthar, M. Matrix type drug delivery system: A review. J. Pharm. Sci. Bio-Sci. Res. 2011, 1, 143–151. [Google Scholar]
- Gavasane, A.J.; Pawar, H.A. Synthetic Biodegradable Polymers Used in Controlled Drug Delivery System: An Overview. Clin. Pharmacol. Biopharm. 2014, 3, 1–7. [Google Scholar] [CrossRef]
- Siegel, R.A.; Rathbone, M.J. Overview of controlled release mechanisms. In Fundamentals and Applications of Controlled Release Drug Delivery; Siepmann, J., Siegel, R., Rathbone, M., Eds.; Springer: Boston, MA, USA, 2012; Chapter 2; pp. 19–43. [Google Scholar]
- Bruschi, M.L. Main mechanisms to control the drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Bruschi, M.L., Ed.; Elsevier: Cambridge, UK, 2015; Chapter 4; pp. 37–62. [Google Scholar]
- Lin, C.-C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef] [PubMed]
- Liechty, W.B.; Kryscio, D.R.; Slaughter, B.V.; Peppas, N.A. Polymers for drug delivery systems. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 149–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocha-García, D.; Guerra-Contreras, A.; Rosales-Mendoza, S.; Palestino, G. Role of porous silicon/hydrogel composites on drug delivery. Open Mater. Sci. 2016, 3, 93–101. [Google Scholar] [CrossRef] [Green Version]
- Narayanaswamy, R.; Torchilin, V.P. Hydrogels and Their Applications in Targeted Drug Delivery. Molecules 2019, 24, 603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Mooney, D.J. Designing hydrogels for controlled drug delivery. Nat. Rev. Mater. 2016, 1, 1–17. [Google Scholar] [CrossRef]
- Sezer, S.; Şahin, İ.; Öztürk, K.; Şanko, V.; Koçer, Z.; Sezer, U.A. Cellulose-based hydrogels as biomaterials. In Cellulose-Based Superabsorbent Hydrogels, 1st ed.; Mondal, M.I.H., Ed.; Springer International Publishing AG: Basel, Switzerland, 2018; Chapter 39; pp. 1–27. [Google Scholar]
- Barman, A.; Das, M. Cellulose-based hydrogels for pharmaceutical and biomedical applications. In Cellulose-Based Superabsorbent Hydrogels, 1st ed.; Mondal, M.I.H., Ed.; Springer International Publishing AG: Basel, Switzerland, 2018; Chapter 36; pp. 1–28. [Google Scholar]
- Kayra, N.; Aytekin, A.Ö. Synthesis of cellulose-based hydrogels: Preparation, formation, mixture, and modification. In Cellulose-Based Superabsorbent Hydrogels, 1st ed.; Mondal, M.I.H., Ed.; Springer International Publishing: Basel, Switzerland, 2018; Chapter 14; pp. 1–28. [Google Scholar]
- Rusu, D.; Ciolacu, D.; Simionescu, B.C. Cellulose-Based hydrogels in tissue engineering applications. Cellul. Chem. Technol. 2019, 53, 907–923. [Google Scholar] [CrossRef]
- Jeong, B.; Kim, S.W.; Bae, Y.H. Thermosensitive sol–gel reversible hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 154–162. [Google Scholar] [CrossRef]
- Marques-Marinho, F.D.; Vianna-Soares, C.D. Cellulose and its derivatives use in the pharmaceutical compounding practice. In Cellulose—Medical, Pharmaceutical and Electronic Applications; Van De Ven, T.G.M., Ed.; Intechopen: London, UK, 2013; Chapter 8; pp. 141–162. [Google Scholar]
- Debele, T.A.; Mekuria, S.L.; Tsai, H.-C. Polysaccharide based nanogels in the drug delivery system: Application as the carrier of pharmaceutical agents. Mater. Sci. Eng. C 2016, 68, 964–981. [Google Scholar] [CrossRef]
- Tayeb, A.H.; Amini, E.; Ghasemi, S.; Tajvidi, M. Cellulose Nanomaterials—Binding Properties and Applications: A Review. Molecules 2018, 23, 2684. [Google Scholar] [CrossRef] [Green Version]
- Ciolacu, D.; Popa, V.I. Nanocelluloses: Preparations, properties, and applications in medicine. In Pulp Production and Processing, 2nd ed.; Popa, V.I., Ed.; Walter de Gruyter GmbH: Berlin/Munich, Germany; Boston, MA, USA, 2020; Chapter 11; pp. 317–340. [Google Scholar]
- Plackett, D.; Letchford, K.; Jackson, J.; Burt, H. A review of nanocellulose as a novel vehicle for drug delivery. Nord. Pulp Pap. Res. J. 2014, 29, 105–118. [Google Scholar] [CrossRef]
- Del Valle, L.J.; Díaz, A.; Puiggalí, J. Hydrogels for Biomedical Applications: Cellulose, Chitosan, and Protein/Peptide Derivatives. Gels 2017, 3, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aravamudhan, A.; Ramos, D.M.; Nada, A.A.; Kumbar, S.G. Natural polymers: Polysaccharides and their derivatives for biomedical applications. In Natural and Synthetic Biomedical Polymers, 1st ed.; Kumbar, S.G., Laurencin, C.T., Deng, M., Eds.; Elsevier: San Diego, CA, USA, 2014; pp. 67–89. [Google Scholar]
- Abeer, M.M.; Amin, M.C.I.M.; Martin, C. A review of bacterial cellulose-based drug delivery systems: Their biochemistry, current approaches and future prospects. J. Pharm. Pharmacol. 2014, 66, 1047–1061. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Rong, L.; Wang, B.; Xie, R.; Sui, X.; Xu, H.; Zhang, L.; Zhong, Y.; Mao, Z. Facile fabrication of redox/pH dual stimuli responsive cellulose hydrogel. Carbohydr. Polym. 2017, 176, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.-H.; Qi, C.; Ma, M.-G.; Wan, P. Multifunctional cellulose-based hydrogels for biomedical applications. J. Mater. Chem. B 2019, 7, 1541–1562. [Google Scholar] [CrossRef] [PubMed]
- Ciolacu, D.; Suflet, M.D. Cellulose-based hydrogels for medical/pharmaceutical applications. In Biomass as Renewable Raw Material to Obtain Bioproducts of High-Tech Value; Popa, V., Volf, I., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 11; pp. 401–439. [Google Scholar]
- Ciolacu, D.; Rudaz, C.; Vasilescu, M.; Budtova, T. Physically and chemically cross-linked cellulose cryogels: Structure, properties and application for controlled release. Carbohydr. Polym. 2016, 151, 392–400. [Google Scholar] [CrossRef]
- Ciolacu, D. Structure-property relationships. In Cellulose-Based Hydrogels; Mondal, M.I.H., Ed.; Springer International Publishing AG: Basel, Switzerland, 2018; Chapter 3; pp. 1–32. [Google Scholar]
- Javanbakht, S.; Shaabani, A. Carboxymethyl cellulose-based oral delivery systems. Int. J. Biol. Macromol. 2019, 133, 21–29. [Google Scholar] [CrossRef]
- Verma, A.; Dubey, J.; Verma, N.; Nayak, A.K. Chitosan-hydroxypropyl methylcellulose matrices as carriers for hydrodynamically balanced capsules of moxifloxacin HCl. Curr. Drug Deliv. 2016, 14, 83–90. [Google Scholar] [CrossRef]
- Majumder, T.; Biswas, G.R.; Majee, S.B. Hydroxy Propyl Methyl Cellulose: Different Aspects in Drug Delivery. J. Pharm. Pharmacol. 2016, 4, 381–385. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Chen, L.; Deng, M.; Bai, Y.; Chen, X.; Jing, X. Biodegradable thermo- and pH-responsive hydrogels for oral drug delivery. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 2941–2951. [Google Scholar] [CrossRef]
- Hasan, A.M.A.; El-Sayed Abdel-Raouf, M. Cellulose-Based Superabsorbent Hydrogels. In Cellulose-Based Superabsorbent Hydrogels, Polymers and Polymeric Composites: A Reference Series; Mondal, M.I.H., Ed.; Springer International Publishing AG: Basel, Swizterland, 2018; pp. 245–267. [Google Scholar]
- Chyzy, A.; Tomczykowa, M.; Plonska-Brzezinska, M.E. Hydrogels as Potential Nano-, Micro- and Macro-Scale Systems for Controlled Drug Delivery. Materials 2020, 13, 188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chatterjee, S.; Hui, P.C.-L. Review of Stimuli-Responsive Polymers in Drug Delivery and Textile Application. Molecules 2019, 24, 2547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshida, R.; Sakai, K.; Okano, T.; Sakurai, Y. Pulsatile drug delivery systems using hydrogels. Adv. Drug Deliv. Rev. 1993, 11, 85–108. [Google Scholar] [CrossRef]
- Caló, E.; Khutoryanskiy, V.V. Biomedical applications of hydrogels: A review of patents and commercial products. Eur. Polym. J. 2015, 65, 252–267. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, S.; Hui, P.C.L. Stimuli-responsive hydrogels: An interdisciplinary overview. In Hydrogels—Smart Materials for Biomedical Applications; Popa, L., Ghica, M.V., Dinu-Pîrvu, C.E., Eds.; IntechOpen Limited: London, UK, 2018; Chapter 2; pp. 1–23. [Google Scholar]
- Wei, M.; Gao, Y.; Li, X.; Serpe, M.J. Stimuli-responsive polymers and their applications. Polym. Chem. 2017, 8, 127–143. [Google Scholar] [CrossRef] [Green Version]
- Onaciu, A.; Munteanu, R.A.; Moldovan, C.S.; Berindan-Neagoe, I. Hydrogels Based Drug Delivery Synthesis, Characterization and Administration. Pharmaceutics 2019, 11, 432. [Google Scholar] [CrossRef] [Green Version]
- Caicco, M.J.; Cooke, M.J.; Wang, Y.; Tuladhar, A.; Morshead, C.M.; Shoichet, M.S. A hydrogel composite system for sustained epi-cortical delivery of Cyclosporin A to the brain for treatment of stroke. J. Control. Release 2013, 166, 197–202. [Google Scholar] [CrossRef]
- Wang, Y.; Cooke, M.J.; Morshead, C.M.; Shoichet, M.S. Hydrogel delivery of erythropoietin to the brain for endogenous stem cell stimulation after stroke injury. Biomaterials 2012, 33, 2681–2692. [Google Scholar] [CrossRef]
- Sood, S.; Gupta, V.K.; Agarwal, S.; Dev, K.; Pathania, D. Controlled release of antibiotic amoxicillin drug using carboxymethyl cellulose-cl-poly(lactic acid-co-itaconic acid) hydrogel. Int. J. Biol. Macromol. 2017, 101, 612–620. [Google Scholar] [CrossRef]
- Perioli, L.; Ambrogi, V.; Rubini, D.; Giovagnoli, S.; Ricci, M.; Blasi, P.; Rossi, C. Novel mucoadhesive buccal formulation containing metronidazole for the treatment of periodontal disease. J. Control. Release 2004, 95, 521–533. [Google Scholar] [CrossRef]
- Fang, J.-Y.; Leu, Y.-L.; Wang, Y.-Y.; Tsai, Y.-H. In vitro topical application and in vivo pharmacodynamic evaluation of nonivamide hydrogels using Wistar rat as an animal model. Eur. J. Pharm. Sci. 2002, 15, 417–423. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Huang, K.-Y.; Yang, B.-Y.; Ko, C.-H.; Huang, H.-M. Fabrication of Novel Hydrogel with Berberine-Enriched Carboxymethylcellulose and Hyaluronic Acid as an Anti-Inflammatory Barrier Membrane. BioMed Res. Int. 2016, 2016, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Moreira, A.P.D.; Thiré, R.M.D.S.M.; Quilty, B.; Passos, T.M.; Simon, P.; Mancini, M.C.; McGuinness, G.B. Absorbent polyvinyl alcohol-sodium carboxymethyl cellulose hydrogels for propolis delivery in wound healing applications. Polym. Eng. Sci. 2017, 57, 1224–1233. [Google Scholar] [CrossRef]
- Sun, N.; Wang, T.; Yan, X. Self-assembled supermolecular hydrogel based on hydroxyethyl cellulose: Formation, in vitro release and bacteriostasis application. Carbohydr. Polym. 2017, 172, 49–59. [Google Scholar] [CrossRef]
- Kong, B.J.; Kim, A.; Park, S.N. Properties and in vitro drug release of hyaluronic acid-hydroxyethyl cellulose hydrogels for transdermal delivery of isoliquiritigenin. Carbohydr. Polym. 2016, 147, 473–481. [Google Scholar] [CrossRef]
- Sawant, P.D.; Luu, D.; Ye, R.; Buchta, R. Drug release from hydroethanolic gels. Effect of drug’s lipophilicity (logP), polymer–drug interactions and solvent lipophilicity. Int. J. Pharm. 2010, 396, 45–52. [Google Scholar] [CrossRef]
- Prakash, P.R.; Raghavendra Rao, N.G.; Soujanya, C. Formulation, evaluation and antiinflammatory activity of topical etoricoxib gel. Asian J. Pharm. Clin. Res. 2010, 3, 126–129. [Google Scholar]
- Bidkar, S.; Devendra, J.; Padsalg, A.; Patel, K.; Mokale, V. Formulation development and evaluation of fluconazole gel in various polymer bases formulation development and evaluation of fluconazole gel in various polymer bases. Asian J. Pharm. 2007, 1, 63–68. [Google Scholar]
- Cho, C.-W.; Choi, J.-S.; Shin, S.-C. Enhanced local anesthetic action of mepivacaine from the bioadhesive gels. Pak. J. Pharm. Sci. 2011, 24, 87–93. [Google Scholar]
- Vlaia, L.; Olariu, I.; Coneac, G.; Muţ, A.M.; Vlaia, V.; Szabadai, Z.; Lupuleasa, D. Formulation, preparation and evaluation of HPMC-based hydroethanolic gels containing propranolol hydrochloride 3% and terpenes. Pract. Farm. 2015, 8, 5–15. [Google Scholar]
- Peppas, N.A. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Oprea, A.-M.; Nistor, M.T.; Popa, M.I.; Lupusoru, C.E.; Vasile, C. In vitro and in vivo theophylline release from cellulose/chondroitin sulfate hydrogels. Carbohydr. Polym. 2012, 90, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Oprea, A.M.; Ciolacu, D.; Neamtu, A.; Mungiu, O.C.; Stoica, B.; Vasile, C. Cellulose/chondroitin sulfate hydrogels: Synthesis, drug loading/release properties and biocompatibility. Cell Chem. Technol. 2010, 44, 369–378. [Google Scholar]
- Baus, R.A.; Zahir-Jouzdani, F.; Dünnhaupt, S.; Atyabi, F.; Bernkop-Schnürch, A. Mucoadhesive hydrogels for buccal drug delivery: In vitro-in vivo correlation study. Eur. J. Pharm. Biopharm. 2019, 142, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Cao, Y.; Song, X.; Zhang, Z.; Zhuang, X.; He, C.; Chen, X. Biodegradable, pH-Responsive Carboxymethyl Cellulose/Poly(Acrylic Acid) Hydrogels for Oral Insulin Delivery. Macromol. Biosci. 2013, 14, 565–575. [Google Scholar] [CrossRef] [PubMed]
- Coviello, T.; Matricardi, P.; Marianecci, C.; Alhaique, F. Polysaccharide hydrogels for modified release formulations. J. Control. Release 2007, 119, 5–24. [Google Scholar] [CrossRef]
- Mawazi, S.M.; Al-Mahmood, S.M.A.; Chatterjee, B.; Hadi, H.A.; Doolaanea, A.A.; Mahmood, A. Hadi Carbamazepine Gel Formulation as a Sustained Release Epilepsy Medication for Pediatric Use. Pharmaceutics 2019, 11, 488. [Google Scholar] [CrossRef] [Green Version]
- Amin, M.C.I.M.; Ahmad, N.; Pandey, M.; Xin, C.J. Stimuli-responsive bacterial cellulose-g-poly(acrylic acid-co-acrylamide) hydrogels for oral controlled release drug delivery. Drug Dev. Ind. Pharm. 2013, 40, 1340–1349. [Google Scholar] [CrossRef]
- Ahmad, N.; Amin, M.C.I.M.; Mahali, S.M.; Ismail, I.; Chuang, V.T.G. Biocompatible and Mucoadhesive Bacterial Cellulose-g-Poly (acrylic acid) Hydrogels for Oral Protein Delivery. Mol. Pharm. 2014, 11, 4130–4142. [Google Scholar] [CrossRef]
- Shi, X.; Zheng, Y.; Wang, G.; Lin, Q.; Fan, J. pH- and electro-response characteristics of bacterial cellulose nanofiber/sodium alginate hybrid hydrogels for dual controlled drug delivery. RSC Adv. 2014, 4, 47056–47065. [Google Scholar] [CrossRef]
- Javanbakht, S.; Pooresmaeil, M.; Hashemi, H.; Namazi, H. Carboxymethylcellulose capsulated Cu-based metal-organic framework-drug nanohybrid as a pH-sensitive nanocomposite for ibuprofen oral delivery. Int. J. Biol. Macromol. 2018, 119, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Salerno, C.; Carlucci, A.M.; Bregni, C. Study of In Vitro Drug Release and Percutaneous Absorption of Fluconazole from Topical Dosage Forms. AAPS PharmSciTech 2010, 11, 986–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abd-Allah, F.I.; Dawaba, H.M.; Ahmed, A.M. Preparation, characterization, and stability studies of piroxicam-loaded microemulsions in topical formulations. Drug Discov. Ther. 2010, 4, 267–275. [Google Scholar] [PubMed]
- Malik, N.S.; Ahmad, M.; Minhas, M.U. Cross-linked β-cyclodextrin and carboxymethyl cellulose hydrogels for controlled drug delivery of acyclovir. PLoS ONE 2017, 12, e0172727. [Google Scholar] [CrossRef] [Green Version]
- Baron, R.I.; Culica, M.E.; Biliuta, G.; Bercea, M.; Gherman, S.; Zavastin, D.; Ochiuz, L.; Avadanei, M.; Coseri, S. Physical Hydrogels of Oxidized Polysaccharides and Poly(Vinyl Alcohol) for Wound Dressing Applications. Materials 2019, 12, 1569. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, G.; Orasugh, J.T.; Saha, N.R.; Roy, I.; Bhattacharyya, A.; Chattopadhyay, A.K.; Rana, D.; Chattopadhyay, D. Cellulose nanofibrils/chitosan based transdermal drug delivery vehicle for controlled release of ketorolac tromethamine. New J. Chem. 2017, 41, 15312–15319. [Google Scholar] [CrossRef]
- Tavakoli, N.; Minaiyan, M.; Heshmatipour, M.; Musavinasab, R. Transdermal iontophoretic delivery of celecoxib from gel formulation. Res. Pharm. Sci. 2016, 10, 419–428. [Google Scholar]
- Nair, A.B.; Vyas, H.; Shah, J.; Kumar, A. Effect of permeation enhancers on the iontophoretic transport of metoprolol tartrate and the drug retention in skin. Drug Deliv. 2010, 18, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Kwon, S.S.; Kong, B.J.; Park, S.N. Physicochemical properties of pH-sensitive hydrogels based on hydroxyethyl cellulose–hyaluronic acid and for applications as transdermal delivery systems for skin lesions. Eur. J. Pharm. Biopharm. 2015, 92, 146–154. [Google Scholar] [CrossRef]
- Trombino, S.; Servidio, C.; Curcio, F.; Cassano, R. Strategies for Hyaluronic Acid-Based Hydrogel Design in Drug Delivery. Pharmaceutics 2019, 11, 407. [Google Scholar] [CrossRef] [Green Version]
- Nandy, B.C.; Gupta, R.N.; Rai, V.K.; Das, S.; Tyagi, L.K.; Roy, S.; Meena, K.C. Transdermal iontophoretic delivery of atenolol in combination with penetration enhancers: Optimization and evaluation on solution and gels. Int. J. Pharmac. Sci. Drug Res. 2009, 1, 91–99. [Google Scholar]
- Alvarez-Lorenzo, C.; Blanco-Fernandez, B.; Puga, A.M.; Concheiro, A. Crosslinked ionic polysaccharides for stimuli-sensitive drug delivery. Adv. Drug Deliv. Rev. 2013, 65, 1148–1171. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.-Y.; Hui, P.C.-L.; Wat, E.; Ng, F.S.F.; Kan, C.-W.; Lau, C.B.-S.; Leung, P.C. Enhanced Transdermal Permeability via Constructing the Porous Structure of Poloxamer-Based Hydrogel. Polymers 2016, 8, 406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De La Fuente, M.; Raviña, M.; Paolicelli, P.; Sanchez, A.; Seijo, B.; Alonso, M.J. Chitosan-based nanostructures: A delivery platform for ocular therapeutics. Adv. Drug Deliv. Rev. 2010, 62, 100–117. [Google Scholar] [CrossRef] [PubMed]
- Madan, M.; Bajaj, A.; Lewis, S.; Udupa, N.; Baig, J.A. In situ forming polymeric drug delivery systems. Indian J. Pharm. Sci. 2009, 71, 242–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubashynskaya, N.V.; Poshina, D.; Raik, S.; Urtti, A.; Skorik, Y.A. Polysaccharides in Ocular Drug Delivery. Pharmaceutics 2019, 12, 22. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Liu, Y.; Li, X.; Kebebe, D.; Zhang, B.; Ren, J.; Lu, J.; Li, J.; Du, S.-Y.; Liu, Z. Research progress of in-situ gelling ophthalmic drug delivery system. Asian J. Pharm. Sci. 2019, 14, 1–15. [Google Scholar] [CrossRef]
- Khan, N.; Aqil, M.; Imam, S.S.; Asgar, A. Development and evaluation of a novel in situ gel of sparfloxacin for sustained ocular drug delivery: In vitro and ex vivo characterization. Pharm. Dev. Technol. 2015, 20, 662–669. [Google Scholar] [CrossRef]
- Duan, X.; Mao, S. New strategies to improve the intranasal absorption of insulin. Drug Discov. Today 2010, 15, 416–427. [Google Scholar] [CrossRef]
- Mawad, D.; Boughton, E.A.; Boughton, P.; Lauto, A. Advances in hydrogels applied to degenerative diseases. Curr. Pharm. Des. 2012, 18, 2558–2575. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Jayakrishnan, A. Injectable hydrogels for biomedical applications. In Injectable Hydrogels for Regenerative Engineering, 2nd ed.; Nair, L.S., Ed.; Imperial College Press: Hackensack, NJ, USA, 2016; pp. 33–96. [Google Scholar]
- Nirmal, H.B.; Bakliwal, S.R.; Pawar, S.P. In-situ gel: New trends in controlled and sustained drug delivery system. Int. J. Pharm. Tech. Res. 2010, 2, 1398–1408. [Google Scholar]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target. Ther. 2018, 3, 1–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, L.; Wang, Q.; Shen, M.; Sun, Y.; Zhang, X.; Huang, C.; Chen, J.; Li, R.; Duan, Y.R. Thermoresponsive nanocomposite gel for local drug delivery to suppress the growth of glioma by inducing autophagy. Autophagy 2017, 13, 1176–1190. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Oh, J.K. Intracellular delivery cellulose-based bionanogels with dual temperature/pH-response for cancer therapy. Colloids Surf. B Biointerfaces 2015, 133, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.N.; Mohan, Y.M.; Varaprasad, K.; Ravindra, S.; Joy, P.A.; Raju, K.M. Magnetic and electric responsive hydrogel-magnetic nanocomposites for drug-delivery application. J. Appl. Polym. Sci. 2011, 122, 1364–1375. [Google Scholar] [CrossRef]
- Oh, J.K.; Drumright, R.; Siegwart, D.J.; Matyjaszewski, K. The development of microgels/nanogels for drug delivery applications. Prog. Polym. Sci. 2008, 33, 448–477. [Google Scholar] [CrossRef]
- Daniel-da-Silva, A.L.; Ferreira, L.; Gil, A.M.; Trindade, T. Synthesis and swelling behavior of temperature responsive k-carrageenan nanogels. J. Colloid Interf. Sci. 2011, 355, 512–517. [Google Scholar] [CrossRef]
- Hajebi, S.; Rabiee, N.; Bagherzadeh, M.; Ahmadi, S.; Rabiee, M.; Roghani-Mamaqani, H.; Tahriri, M.; Taheby, L.; Hamblin, M.R. Stimulus-responsive polymeric nanogels as smart drug delivery systems. Acta Biomater. 2019, 92, 1–18. [Google Scholar] [CrossRef]
- Chang, C.; Zhang, L. Cellulose-based hydrogels: Present status and application prospects. Carbohydr. Polym. 2011, 84, 40–53. [Google Scholar] [CrossRef]
- Xing, C.; Chen, S.; Qiu, M.; Liang, X.; Liu, Q.; Zou, Q.; Li, Z.; Xie, Z.; Wang, D.; Dong, B.; et al. Conceptually Novel Black Phosphorus/Cellulose Hydrogels as Promising Photothermal Agents for Effective Cancer Therapy. Adv. Health Mater. 2018, 7, e1701510. [Google Scholar] [CrossRef]
- Javanbakht, S.; Namazi, H. Doxorubicin loaded carboxymethyl cellulose/graphene quantum dot nanocomposite hydrogel films as a potential anticancer drug delivery system. Mater. Sci. Eng. C 2018, 87, 50–59. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Liang, H.; Lin, L.; Shah, B.R.; Li, Y.; Chen, Y.; Li, B. Green-step assembly of low density lipoprotein/sodium carboxymethyl cellulose nanogels for facile loading and pH-dependent release of doxorubicin. Colloids Surf. B Biointerfaces 2015, 126, 288–296. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Wang, X.; Yuan, K.; Xie, C.; Wu, W.; Jiang, X.; Hu, L. Delivery of doxorubicin in vitro and in vivo using bio-reductive cellulose nanogels. Biomater. Sci. 2014, 2, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Won, Y.-W.; Lim, K.S.; Kim, Y.-H. Low-Molecular-Weight Methylcellulose-Based Thermo-reversible Gel/Pluronic Micelle Combination System for Local and Sustained Docetaxel Delivery. Pharm. Res. 2011, 29, 525–534. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, R.K. Use of Polysaccharide Hydrogels in Drug Delivery and Tissue Engineering. Adv. Tissue Eng. Regen. Med. Open Access 2017, 2, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Mathew, A.P.; Uthaman, S.; Cho, K.; Cho, C.-S.; Park, I.-K. Injectable hydrogels for delivering biotherapeutic molecules. Int. J. Biol. Macromol. 2018, 110, 17–29. [Google Scholar] [CrossRef]
- Thambi, T.; Phan, V.H.G.; Lee, D.S. Stimuli-Sensitive Injectable Hydrogels Based on Polysaccharides and Their Biomedical Applications. Macromol. Rapid Commun. 2016, 37, 1881–1896. [Google Scholar] [CrossRef]
- Lv, S.; Liu, M.; Ni, B.; Lv, S. An injectable oxidized carboxymethylcellulose/N-succinyl-chitosan hydrogel system for protein delivery. Chem. Eng. J. 2010, 160, 779–787. [Google Scholar] [CrossRef]















| Location | pH | |||
|---|---|---|---|---|
| Average pH [16] | Children [17] | Adults [17] | ||
| Oral cavity | 5.8–7.4 | - | - | |
| Esophagus | 5.0–6.0 | - | - | |
| Stomach | Fasted conditions | 1.5–2.0 | 1.5 | 1–2.5 |
| Fed conditions | 3.0–5.0 | - | - | |
| Small intestine | Duodenum | - | 6.3–6.4 | 5–6.5 |
| Jejunum | 5.0–6.5 | 6.6–7 | 5.8–7.7 | |
| Ileum | 6.0–7.5 | 7.3 | 6.6–7.7 | |
| Large intestine | Cecum | - | 5.8–5.9 | 6–6.5 |
| Ascending colon (right colon) | 6.4 | 5.9–6.5 | 4.6–6.8 | |
| Transverse colon (mid colon) | 6.0–7.6 | 5.3 | 4.6–7.1 | |
| Descending colon (left colon) | 6.0–7.6 | 6 | 5.5–7.4 | |
| Rectum | - | 6.5 | 5.3–7.4 | |
| Cellulose Derivatives | Drug | Treatment | Ref. |
|---|---|---|---|
| Methylcellulose (MC) | Cyclosporine A | Sustained brain delivery | [84] |
| Erythropoietin | Delivery to the brain in order to endogenous stem cell stimulation after stroke | [85] | |
| Carboxymethylcellulose (CMC) | Amoxicillin | Excellent antibacterial agent against gram-positive Staphylococcus aureus | [86] |
| Acyclovir | Controlled drug delivery systems | [87] | |
| Diclofenac | Skin wounds | [1] | |
| Nonivamide | Improved skin permeation and distribution | [88] | |
| Berberine | Protect postsurgical tissue and perform a controlled drug release | [89] | |
| Propolis | Wound healing | [90] | |
| Hydroxyethyl cellulose (HEC) | Eugenol | Efficient bacteriostasis against Escherichia coli | [91] |
| Isoliquiritigenin | Transdermal delivery system | [92] | |
| Hydroxypropyl cellulose (HPC) | Lidocaine | Promote a systematical and controlled drug | [93] |
| Hydroxypropyl methylcellulose (HPMC) | Etoricoxib | Chronic or acute illness | [94] |
| Fluconazole | Skin fungal infections | [95] | |
| Mepivacaine | Relieve local pain and perform a controlled drug release | [96] | |
| Propranolol | Improve percutaneous penetration | [97] |
| Cellulose-Based Hydrogels | Physical Properties | Drug Release Efficiency | Ref. |
|---|---|---|---|
| CMC dual temperature/pH response nanogels (DuR-BNGs) |
|
| [132] |
| Cellulose/BPNSs nanocomposite hydrogels |
|
| [138] |
| CMC/GQDs nanocomposite hydrogels |
|
| [139] |
| LDL/CMC nanogels |
|
| [140] |
| Methacrylated/ CMC nanogels |
|
| [141] |
| LMw MC/ Pluronic F127 hydrogels |
|
| [142] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciolacu, D.E.; Nicu, R.; Ciolacu, F. Cellulose-Based Hydrogels as Sustained Drug-Delivery Systems. Materials 2020, 13, 5270. https://0-doi-org.brum.beds.ac.uk/10.3390/ma13225270
Ciolacu DE, Nicu R, Ciolacu F. Cellulose-Based Hydrogels as Sustained Drug-Delivery Systems. Materials. 2020; 13(22):5270. https://0-doi-org.brum.beds.ac.uk/10.3390/ma13225270
Chicago/Turabian StyleCiolacu, Diana Elena, Raluca Nicu, and Florin Ciolacu. 2020. "Cellulose-Based Hydrogels as Sustained Drug-Delivery Systems" Materials 13, no. 22: 5270. https://0-doi-org.brum.beds.ac.uk/10.3390/ma13225270