Tablet of Spondias mombin L. Developed from Nebulized Extract Prevents Gastric Ulcers in Mice via Cytoprotective and Antisecretory Effects
Abstract
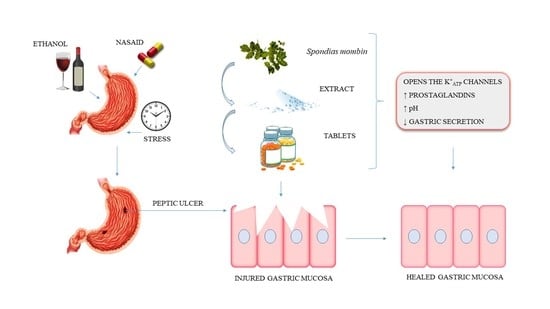
:1. Introduction
2. Materials and Methods
2.1. Plant and Extract Preparation
2.2. Phytochemical Screening
2.3. Identification of Chemical Markers by High-Performance Liquid Chromatography (HPLC)
2.4. Animals
2.5. Acute Toxicity Study
2.6. Evaluation of Gastroprotective Activity and Possible Mechanisms of Action of SmNE
2.6.1. Ethanol-Induced Ulcers
2.6.2. Ulcers Induced by Nonsteroidal Anti-Inflammatory (Piroxicam)
2.6.3. Determination of the Ulcerative Lesion Index
- Score 1: hemorrhagic stitches and ulcerations up to 1 mm.
- Score 2: ulcerations of 2 mm.
- Score 3: deep ulcerations of 3 mm or more.
2.6.4. Determination of Gastric Acid Secretion
2.6.5. Assessment of the Involvement of Prostaglandins (PGs)
2.6.6. Assessment of the Involvement of ATP-Sensitive Potassium Channels (KATP)
2.7. Development of the Tablets
2.7.1. Technological Parameters
2.7.2. Rest Angle
2.7.3. Production of Tablets
2.8. Tablet Quality Parameters
2.8.1. Average Weight
2.8.2. Hardness
2.8.3. Friability
2.8.4. Disintegration
2.9. Evaluation of the Gastroprotective Activity of the Tablet Formulation by the Ethanol-Induced Ulcer Model
2.10. Statistical Analysis
3. Results and Discussion
3.1. Phytochemical Screening
3.2. High-Performance Liquid Chromatography (HPLC)
3.3. Acute Toxicity
3.4. Ethanol-Induced Ulcers
3.5. Ulcers Induced by Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
3.6. Evaluation of Antisecretory Activity
3.7. Evaluation of the Role of Prostaglandins (PG’s) in the Gastroprotective Effect
3.8. Evaluation Involving ATP-Sensitive Potassium Channels in the Gastroprotective Effect
3.9. Formulation Development
3.10. Evaluation of the Quality Parameters of the Pills (P2)
3.10.1. Determination of the Average Weight of the Tablets
3.10.2. Determination of Hardness
3.10.3. Assessment of Friability
3.10.4. Determination of Disintegration Time
3.11. Evaluation of the Gastroprotective Activity of the Tablet Formulation in the Ethanol-Induced Ulcer Model
4. Conclusions
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Lanas, A.; Chan, F. Peptic ulcer disease. Lancet 2017, 390, 613–624. [Google Scholar] [CrossRef]
- Byun, S.H.; Min, C.; Hong, S.J.; Choi, H.G.; Koh, D.H. Analysis of the relation between periodontitis and chronic gastritis/peptic ulcer: A cross-sectional study using KoGES HEXA data. Int. J. Environ. Res. Public Health 2020, 17, 4387. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.C.; Chen, T.P.; Wei, C.Y.; Chang, Y.C. Association between peptic ulcer disease and periodontitis: A nationwide population-based case-control study in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, A.R.S.; Diniz, P.B.F.; Pinheiro, M.S.; Albuquerque-Júnior, R.L.C.; Thomazzi, S.M. Gastroprotective effects of thymol on acute and chronic ulcers in rats: The role of prostaglandins, ATP-sensitive K+ channels, and gastric mucus secretion. Chem. Biol. Interact. 2016, 244, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.K.; Choi, M.G.; Choi, S.C.; Lee, K.M.; Kim, T.O.; Park, S.H.; Moon, J.S.; Lim, Y.J.; Kang, D.H.; Cheon, G.J.; et al. Randomised clinical trial: Tegoprazan, a novel potassium-competitive acid blocker, or lansoprazole in the treatment of gastric ulcer. Aliment. Pharmacol. Ther. 2020, 52, 789–797. [Google Scholar] [CrossRef]
- Bansal, V.K.; Goel, R.K. Gastroprotective effect of Acacia nilotica young seedless pod extract: Role of polyphenolic constituents. Asian Pac. J. Trop. Med. 2012, 5, 523–528. [Google Scholar] [CrossRef]
- Vaezi, M.F.; Yang, Y.X.; Howden, C.W. Complications of Proton Pump Inhibitor Therapy. Gastroenterology 2017, 153, 35–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Q.; Wong, C.H.; Chan, H.Y.E.; Lee, W.Y.; Zuo, Z. Statistical Design of Experiment (DoE) based development and optimization of DB213 in situ thermosensitive gel for intranasal delivery. Int. J. Pharm. 2018, 539, 50–57. [Google Scholar] [CrossRef]
- Sabiu, S.; Garuba, T.; Sunmonu, T.O.; Sulyman, A.O.; Ismail, N.O. Indomethacin-induced gastric ulceration in rats: Ameliorative roles of Spondias mombin and Ficus exasperata. Pharm. Biol. 2016, 54, 180–186. [Google Scholar] [CrossRef]
- Escobedo-Hinojosa, W.I.; Gomez-Chang, E.; García-Martínez, K.; Guerrero Alquicira, R.; Cardoso-Taketa, A.; Romero, I. Gastroprotective Mechanism and Ulcer Resolution Effect of Cyrtocarpa procera Methanolic Extract on Ethanol-Induced Gastric Injury. Evid. Based Complement. Altern. Med. 2018, 2018. [Google Scholar] [CrossRef] [Green Version]
- Cristofoli, N.L.; Lima, C.A.R.; Vieira, M.M.C.; Andrade, K.S.; Ferreira, S.R.S. Antioxidant and antimicrobial potential of cajazeira leaves (Spondias mombin) extracts. Sep. Sci. Technol. 2019, 54, 580–590. [Google Scholar] [CrossRef]
- dos Santos Sampaio, T.I.; de Melo, N.C.; de Freitas Paiva, B.T.; da Silva Aleluia, G.A.; da Silva Neto, F.L.P.; da Silva, H.R.; Keita, H.; Cruz, R.A.S.; Sánchez-Ortiz, B.L.; Pineda-Peña, E.A.; et al. Leaves of Spondias mombin L. a traditional anxiolytic and antidepressant: Pharmacological evaluation on zebrafish (Danio rerio). J. Ethnopharmacol. 2018, 224, 563–578. [Google Scholar] [CrossRef] [PubMed]
- Shakya, A.K. Medicinal plants: Future source of new drugs. Int. J. Herb. Med. 2016, 4, 59–64. [Google Scholar]
- Fuenmayor, E.; Forde, M.; Healy, A.V.; Devine, D.M.; Lyons, J.G.; McConville, C.; Major, I. Comparison of fused-filament fabrication to direct compression and injection molding in the manufacture of oral tablets. Int. J. Pharm. 2019, 558, 328–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandra, S.; De Mejia, E.G. Polyphenolic compounds, antioxidant capacity, and quinone reductase activity of an aqueous extract of Ardisia compressa in comparison to mate (llex paraguariensis) and green (Camellia sinensis) teas. J. Agric. Food Chem. 2004, 52, 3583–3589. [Google Scholar] [CrossRef] [PubMed]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activity. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Becker, K. Vanillin-HCl method for condensed tannins: Effect of organic solvents used for extraction of tannins. J. Chem. Ecol. 1993, 19, 613–621. [Google Scholar] [CrossRef]
- OECD. OECD—Guideline for Testing of Chemicals: Acute Oral Toxicity—Acute Toxic Class Method; OECD: Paris, France, 2001; pp. 1–14. [Google Scholar] [CrossRef]
- Almeida, R.N.; Falcão, A.C.G.M.; Diniz, R.S.T.; Quintas-Júnior, L.J.; Polari, R.M.; Barbosa-Filho, J.M.; Agra, M.F.; Duarte, J.C.; Ferreira, C.D.; Antoniol, A.R. Metodologia para avaliação de plantas com atividade no sistema nervoso central e alguns dados experimentais. Rev. Bras. Farm. 1999, 80, 72–76. [Google Scholar]
- Robert, A.; Nezamis, J.E.; Lancaster, C.; Hanchar, A.J. Cytoprotection by prostaglandins in rats. Prevention of gastric necrosis produced by alcohol, HCl, NaOH, hypertonic NaCl, and thermal injury. Gastroenterology 1979, 77, 433–443. [Google Scholar] [CrossRef]
- Puscas, I.; Puscas, C.; Coltau, M.; Pasca, R.; Torres, J.; Márquez, M.; Herrero, E.; Fillat, O.; Ortiz, J.A. Comparative Study of the Safety and Efficacy of Ebrotidine Versus Ranitidine and Placebo in the Prevention of Piroxicam-Induced Gastroduodenal Lesions. Arzneimittelforschung 1997, 47, 568–572. [Google Scholar] [PubMed]
- Szelenyi, I.; Thiemer, K. Distention ulcer as a model for testing of drugs for ulcerogenic side effects. Arch. Toxicol. 1978, 41, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Shay, H.; Komarov, S.A.; Fels, S.S.; Meranze, D.; Gruenstein, M.; Siplet, H. A simple method for the uniform production of gastric ulceration in the rat. Gastroenterology 1945, 5, 43–61. [Google Scholar]
- Adeyemi, E.O.; Bastaki, S.A.; Chandranath, I.S.; Hasan, M.Y.; Fahim, M.; Adem, A. Mechanisms of action of leptin in preventing gastric ulcer. World J. Gastroenterol. 2005, 11, 4154–4160. [Google Scholar] [CrossRef] [PubMed]
- Peskar, B.M.; Ehrlich, K.; Peskar, B.A. Role of ATP-sensitive potassium channels in prostaglandin-mediated gastroprotection in the rat. J. Pharmacol. Exp. Ther. 2002, 301, 969–974. [Google Scholar] [CrossRef]
- Gallo, L.; Llabot, J.M.; Allemandi, D.; Bucalá, V.; Piña, J. Influence of spray-drying operating conditions on Rhamnus purshiana (Cáscara sagrada) extract powder physical properties. Powder Technol. 2011, 208, 205–214. [Google Scholar] [CrossRef]
- Santana, C.P.; Medeiros, F.D.; Correia, L.P.; Diniz, P.H.G.D.; Véras, G.; Medeiros, A.C.D. Dissolution and uniformity of content of tablets developed with extract of Ximenia americana L. PLoS ONE 2018, 13, e0197323. [Google Scholar] [CrossRef]
- Hatcher, R.A. The United States Pharmacopeia; United States Pharmacopoeia Commission: North Bethesda, MD, USA, 2013. [Google Scholar]
- Maduka, H.C.C.; Okpogba, A.N.; Ugwu, C.E.; Dike, C.C.; Ogueche, P.N.; Onwuzurike, D.T.; Ibe, D.C. Phytochemical, antioxidant and microbial inhibitory effects of Spondias mombin leaf and stem bark extracts. IOSR J. Pharm. Biol. Sci. 2014, 9, 14–17. [Google Scholar] [CrossRef]
- Da Silva, A.R.A.; De Morais, S.M.; Mendes Marques, M.M.; De Oliveira, D.F.; Barros, C.C.; De Almeida, R.R.; Vieira, Í.G.P.; Guedes, M.I.F. Chemical composition, antioxidant and antibacterial activities of two Spondias species from Northeastern Brazil. Pharm. Biol. 2012, 50, 740–746. [Google Scholar] [CrossRef]
- Oliveira, O.W.; Petrovick, P.R. Secagem por aspersão (spray drying) de extratos vegetais: Bases e aplicações. Braz. J. Pharmacogn. 2010, 20, 641–650. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, F.H.A.; de Batista, R.S.A.; de Medeiros, F.D.; Santos, F.S.; Medeiros, A.C.D. Development of a rapid and simple HPLC-UV method for determination of gallic acid in Schinopsis brasiliensis. Rev. Bras. Farmacogn. 2015, 25, 208–211. [Google Scholar] [CrossRef] [Green Version]
- Choubey, S.; Goyal, S.; Varughese, L.R.; Kumar, V.; Sharma, A.K.; Beniwal, V. Probing Gallic Acid for Its Broad Spectrum Applications. Mini-Rev. Med. Chem. 2018, 18, 1283–1293. [Google Scholar] [CrossRef]
- Brito, S.A.; Barbosa, I.S.; De Almeida, C.L.F.; De Medeiros, J.W.; Silva Neto, J.C.; Rolim, L.A.; Da Silva, T.G.; Ximenes, R.M.; De Menezes, I.R.A.; Caldas, G.F.R.; et al. Evaluation of gastroprotective and ulcer healing activities of yellow mombin juice from Spondias mombin L. PLoS ONE 2018, 13, e0201561. [Google Scholar] [CrossRef]
- Luiz-Ferreira, A.; De Almeida, A.C.A.; Cola, M.; Barbastefano, V.; De Almeida, A.B.A.; Batista, L.M.; Farias-Silva, E.; Pellizzon, C.H.; Hiruma-Lima, C.A.; Santos, L.C.; et al. Mechanisms of the gastric antiulcerogenic activity of Anacardium humile St. Hil on ethanol-induced acute gastric mucosal injury in rats. Molecules 2010, 15, 7153–7166. [Google Scholar] [CrossRef] [PubMed]
- Abdelwahab, S.I. Protective mechanism of gallic acid and its novel derivative against ethanol-induced gastric ulcerogenesis: Involvement of immunomodulation markers, Hsp70 and Bcl-2-associated X protein. Int. Immunopharmacol. 2013, 16, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Boeing, T.; Da Silva, L.M.; Somensi, L.B.; Cury, B.J.; Michels Costa, A.P.; Petreanu, M.; Niero, R.; De Andrade, S.F. Antiulcer mechanisms of Vernonia condensata Baker: A medicinal plant used in the treatment of gastritis and gastric ulcer. J. Ethnopharmacol. 2016, 184, 196–207. [Google Scholar] [CrossRef]
- Drini, M. Peptic ulcer disease and non-steroidal anti-inflammatory drugs. Aust. Prescr. 2017, 40, 91–93. [Google Scholar] [CrossRef]
- Santenna, C.; Kumar, S.; Balakrishnan, S.; Jhaj, R.; Ahmed, S.N. A comparative experimental study of analgesic activity of a novel non-steroidal anti-inflammatory molecule—Zaltoprofen, and a standard drug—Piroxicam, using murine models. J. Exp. Pharmacol. 2019, 11, 85–91. [Google Scholar] [CrossRef] [Green Version]
- De Olinda, T.M.; Lemos, T.L.G.; Machado, L.L.; Rao, V.S.; Santos, F.A. Quebrachitol-induced gastroprotection against acute gastric lesions: Role of prostaglandins, nitric oxide and K+ATP channels. Phytomedicine 2008, 15, 327–333. [Google Scholar] [CrossRef]
- Toma, W.; Trigo, J.R.; De Paula, A.C.B.; Brito, A.R.M.S. Preventive activity of pyrrolizidine alkaloids from Senecio brasiliensis (Asteraceae) on gastric and duodenal induced ulcer on mice and rats. J. Ethnopharmacol. 2004, 95, 345–351. [Google Scholar] [CrossRef]
- Santos Cerqueira, G.; Dos Santos, E.; Silva, G.; Rios Vasconcelos, E.; Fragoso De Freitas, A.P.; Arcanjo Moura, B.; Silveira MacEdo, D.; Lopes Souto, A.; Barbosa Filho, J.M.; De Almeida Leal, L.K.; et al. Effects of hecogenin and its possible mechanism of action on experimental models of gastric ulcer in mice. Eur. J. Pharmacol. 2012, 683, 260–269. [Google Scholar] [CrossRef] [Green Version]
- Ramadan, M.A.; Shawkey, A.E.; Rabeh, M.A.; Abdellatif, A.O. Promising antimicrobial activities of oil and silver nanoparticles obtained from Melaleuca alternifolia leaves against selected skin-infecting pathogens. J. Herb. Med. 2019, 20, 100289. [Google Scholar] [CrossRef]
- Zakaria, Z.A.; Abdul Hisam, E.E.; Norhafizah, M.; Rofiee, M.S.; Othman, F.; Hasiah, A.H.; Vasudevan, M. Methanol extract of bauhinia purpurea leaf possesses anti-ulcer activity. Med. Princ. Pract. 2012, 21, 476–482. [Google Scholar] [CrossRef]
- El-Hefnawy, M.E.; Alghamdi, Y. Characteristics of ternary clove/water/1-butanol microemulsions based drug delivery system for aspirin. J. Dispers. Sci. Technol. 2019, 40, 1–8. [Google Scholar] [CrossRef]
- Sepulveda, B.; Quispe, C.; Simirgiotis, M.; Torres-Benítez, A.; Reyes-Ortíz, J.; Areche, C.; García-Beltrán, O. Gastroprotective activity of synthetic coumarins: Role of endogenous prostaglandins, nitric oxide, non-protein sulfhydryls and vanilloid receptors. Bioorg. Med. Chem. Lett. 2016, 26, 5732–5735. [Google Scholar] [CrossRef] [PubMed]
- Sousa, G.A.; Oliveira, I.S.; Silva-Freitas, F.V.; Viana, A.F.S.C.; Neto, B.P.S.; Cunha, F.V.M.; Gonçalves, R.L.G.; Lima Filho, A.C.M.; Amaral, M.P.M.; Oliveira, R.d.C.M.; et al. Gastroprotective effect of ethanol extracts of cladodes and roots of Pilosocereus gounellei (A. Weber ex K. Schum.) Bly. Ex Rowl (Cactaceae) on experimental ulcer models. J. Ethnopharmacol. 2018, 218, 100–108. [Google Scholar] [CrossRef]
- Rozza, A.L.; Hiruma-Lima, C.A.; Takahira, R.K.; Padovani, C.R.; Pellizzon, C.H. Effect of menthol in experimentally induced ulcers: Pathways of gastroprotection. Chem. Biol. Interact. 2013, 206, 272–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Yokoyama, W.; Xu, S.; Zhu, S.; Ma, J.; Zhong, F. Formation and stability of W/O microemulsion formed by food grade ingredients and its oral delivery of insulin in mice. J. Funct. Foods 2017, 30, 134–141. [Google Scholar] [CrossRef]
- De Stefani, D. Identification of an ATP-Sensitive Potassium Channel in the Inner Mitochondrial Membrane. Biophys. J. 2020, 118, 1a. [Google Scholar] [CrossRef]
- Medeiros, J.V.R.; Gadelha, G.G.; Lima, S.J.; Garcia, J.A.; Soares, P.M.G.; Santos, A.A.; Brito, G.A.C.; Ribeiro, R.A.; Souza, M.H.L.P. Role of the NO/cGMP/K ATP pathway in the protective effects of sildenafil against ethanol-induced gastric damage in rats. Br. J. Pharmacol. 2008, 153, 721–727. [Google Scholar] [CrossRef] [Green Version]
- Silvéria, S.R.; Rao, V.S.; Carvalho, A.C.S.; Guedes, M.M.; De Morais, T.C.; De Souza, A.L.; Trevisan, M.T.S.; Lima, A.F.; Chaves, M.H.; Santos, F.A. Gastroprotective effect of lupeol on ethanol-induced gastric damage and the underlying mechanism. Inflammopharmacology 2009, 17, 221–228. [Google Scholar] [CrossRef]
- Vidal, C.S.; Oliveira Brito Pereira Bezerra Martins, A.; de Alencar Silva, A.; de Oliveira, M.R.C.; Ribeiro-Filho, J.; de Albuquerque, T.R.; Coutinho, H.D.M.; da Silva Almeida, J.R.G.; Quintans, L.J.; de Menezes, I.R.A. Gastroprotective effect and mechanism of action of Croton rhamnifolioides essential oil in mice. Biomed. Pharmacother. 2017, 89, 47–55. [Google Scholar] [CrossRef]
- Rofaeil, R.R.; Gaber, S.S. Gastroprotective effect of memantine in indomethacin-induced peptic ulcer in rats, a possible role for potassium channels. Life Sci. 2019, 217, 164–168. [Google Scholar] [CrossRef]
- Loch-Neckel, G.; Schütz, F.E.; Derner, R.B.; Lemos-Senna, E. Obtenção de extratos secos de carotenoides a partir da biomassa da microalga Haematococcus pluvialis por secagem em torre de aspersão (spray-drying). Matéria 2018, 23. [Google Scholar] [CrossRef] [Green Version]
- Bushra, R.; Shoaib, M.H.; Aslam, N.; Mehmood, Z.A.; Hashmat, D. Enteric coating of ibuprofen tablets (200 mg) using an aqueous dispersion system. Braz. J. Pharm. Sci. 2010, 46, 99–107. [Google Scholar] [CrossRef]
- Agência Nacional de Vigilância Sanitária—Anvisa. Farmacopeia Brasileira, 6ath ed.; Agência Nacional de Vigilância Sanitária—Anvisa: Brasília, Brazil, 2019; pp. 60–69, 72–85. [Google Scholar] [CrossRef]
- Bersani-Amado, C.A.; Massao, L.B.; Baggio, S.R.; Johanson, L.; Albiero, A.L.M.; Kimura, E. Antiulcer effectiveness of Maytenus aquifolium spray dried extract. Phyther. Res. 2000, 14, 543–545. [Google Scholar] [CrossRef]








| Raw Materials | Function | Formulation 1 | Formulation 2 |
|---|---|---|---|
| Nebulized extract of Spondias mombin (SmNE) | VAPI | 250 mg | 250 mg |
| Microcrystalline Cellulose 101 | Diluent | 155 mg | 452 mg |
| Carboxymethylcellulose | Binder | 10 mg | 10 mg |
| Glycolate starch | Disintegrating | 10 mg | 16 mg |
| Magnesium stearate | Lubricant | 25 mg | 24 mg |
| Talc | Lubricant | 50 mg | 48 mg |
| Treatment (p.o) | pH | Gastric Volume (mL) | Gastric Content (mg) |
|---|---|---|---|
| Saline (10 mL/kg) | 3.3 ± 0.8 | 0.6 ± 0.1 | 593 ± 234. |
| Lansoprazole (30 mg/kg) | 4.7 ± 0.6 ** | 0.12 ± 0.08 ** | 235 ± 78 ** |
| SmNE (250 mg/kg) | 4.5 ± 0.5 * | 0.28 ± 0.08 * | 276 ± 98 * |
| Properties | Formulation 1 | Formulation 2 |
|---|---|---|
| Apparently density (g/cm3) | 0.35 | 0.47 |
| Compaction density (g/cm3) | 0.43 | 0.58 |
| Resting angle (α) | 41.00 | 36.00 |
| Carr Index (CI%) | 25.00 | 20.00 |
| Hausner index (PH) | 1.38 | 1.25 |
| Parameter | Result | Specification |
|---|---|---|
| Average weight (mg) | 804 | 764.10–844.62 |
| Hardness (N) | 35.72 | >30 N |
| Friability (%) | 0.13 | ˂1.5% |
| Disintegration time (m) | 6:15 | <30 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araruna, M.E.; Silva, P.; Almeida, M.; Rêgo, R.; Dantas, R.; Albuquerque, H.; Cabral, I.; Apolinário, N.; Medeiros, F.; Medeiros, A.; et al. Tablet of Spondias mombin L. Developed from Nebulized Extract Prevents Gastric Ulcers in Mice via Cytoprotective and Antisecretory Effects. Molecules 2021, 26, 1581. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules26061581
Araruna ME, Silva P, Almeida M, Rêgo R, Dantas R, Albuquerque H, Cabral I, Apolinário N, Medeiros F, Medeiros A, et al. Tablet of Spondias mombin L. Developed from Nebulized Extract Prevents Gastric Ulcers in Mice via Cytoprotective and Antisecretory Effects. Molecules. 2021; 26(6):1581. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules26061581
Chicago/Turabian StyleAraruna, Maria Elaine, Pablo Silva, Maria Almeida, Renaly Rêgo, Raiff Dantas, Hilton Albuquerque, Ingrid Cabral, Nadjaele Apolinário, Francinalva Medeiros, Ana Medeiros, and et al. 2021. "Tablet of Spondias mombin L. Developed from Nebulized Extract Prevents Gastric Ulcers in Mice via Cytoprotective and Antisecretory Effects" Molecules 26, no. 6: 1581. https://0-doi-org.brum.beds.ac.uk/10.3390/molecules26061581