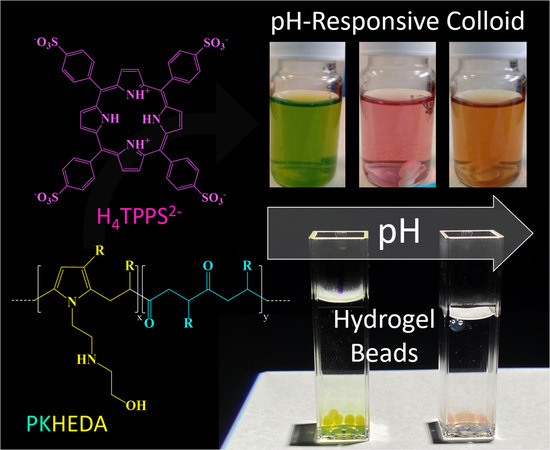
pH-Responsive Polyketone/5,10,15,20-Tetrakis-(Sulfonatophenyl)Porphyrin Supramolecular Submicron Colloidal Structures
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Characterization
2.3. Surface Activity Study of Functional Polyketones
2.4. Solubility Studies of Functional Polyketones
2.5. Study of PKHEDA/TPPS Colloids
2.6. Ultrafiltration
2.7. Colloidal Stability
2.8. Calcium Alginate Sensor Beads
3. Result and Discussion
3.1. Polyketone Modification
3.2. Surface Tension and Self-Aggregation Process of PKHEDA
3.3. PKHEDA/TPPS Colloids
3.4. Ultrafiltration Studies
3.5. Colloid Stability
3.6. Applications
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Lehn, J.M. Cryptates: Inclusion complexes of macropolycyclic receptor molecules. Pure Appl. Chem. 1978, 50, 871–892. [Google Scholar] [CrossRef]
- Albrecht, M. Supramolecular chemistry—General principles and selected examples from anion recognition and metallosupramolecular chemistry. Naturwissenschaften 2007, 94, 951–966. [Google Scholar] [CrossRef] [PubMed]
- Boekhoven, J.; Stupp, S.I. 25th anniversary article: Supramolecular materials for regenerative medicine. Adv. Mater. 2014, 26, 1642–1659. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.; Tang, B.; Liu, Y.; Gao, Q.; Wang, Z.; Zhang, X. The fabrication of a supra-amphiphile for dissipative self-assembly. Chem. Sci. 2016, 7, 1151–1155. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gröhn, F.; Klein, K.; Koynov, K. A novel type of vesicles based on ionic and π–π interactions. Macromol. Rapid Commun. 2010, 31, 75–80. [Google Scholar] [CrossRef]
- Li, Q.; Luo, L.; Yan, X.; Zhou, W.; Wang, F. Ionic self-assembled fluorescent microfibres with electrochemical properties. Supramol. Chem. 2014, 26, 358–362. [Google Scholar] [CrossRef]
- Serpe, M.J.; Craig, S.L. Physical organic chemistry of supramolecular polymers. Langmuir 2007, 23, 1626–1634. [Google Scholar] [CrossRef]
- Lehn, J.-M. Toward complex matter: Supramolecular chemistry and self-organization. Proc. Natl. Acad. Sci. USA 2002, 99, 4763. [Google Scholar] [CrossRef] [Green Version]
- Ruthard, C.; Maskos, M.; Yildiz, H.; Gröhn, F. Association of a cylindrical polyelectrolyte brush with tetravalent counterions. Macromol. Rapid Commun. 2011, 32, 523–527. [Google Scholar] [CrossRef]
- Yan, Y.; Zhao, Y.S. Organic nanophotonics: From controllable assembly of functional molecules to low-dimensional materials with desired photonic properties. Chem. Soc. Rev. 2014, 43, 4325–4340. [Google Scholar] [CrossRef]
- Zhang, Z.; Ma, R.; Shi, L. Cooperative macromolecular self-assembly toward polymeric assemblies with multiple and bioactive functions. Account. Chem. Res. 2014, 47, 1426–1437. [Google Scholar] [CrossRef] [PubMed]
- Shibu, E.S.; Sonoda, A.; Tao, Z.; Feng, Q.; Furube, A.; Masuo, S.; Wang, L.; Tamai, N.; Ishikawa, M.; Biju, V. Energy materials: Supramolecular nanoparticles for solar energy harvesting. Nano Rev. 2013, 4, 21079. [Google Scholar] [CrossRef] [PubMed]
- Hasobe, T.; Murata, H.; Fukuzumi, S.; Kamat, P.V. Porphyrin-based molecular architectures for light energy conversion. Mol. Cryst. Liq. Cryst. 2007, 471, 39–51. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, X.; Wang, R.; Xu, H.; Chi, B. Supramolecular assemblies of histidinylated β-cyclodextrin for enhanced oligopeptide delivery into osteoclast precursors. J. Biomater. Sci. Polym. Ed. 2016, 27, 490–504. [Google Scholar] [CrossRef]
- Frühbeißer, S.; Gröhn, F. Catalytic activity of macroion–porphyrin nanoassemblies. J. Am. Chem. Soc. 2012, 134, 14267–14270. [Google Scholar] [CrossRef]
- Ruthard, C.; Schmidt, M.; Gröhn, F. Porphyrin–polymer networks, worms, and nanorods: pH-triggerable hierarchical self-assembly. Macromol. Rapid Commun. 2011, 32, 706–711. [Google Scholar] [CrossRef]
- Zhao, M.; Zhao, Y.; Zheng, L.; Dai, C. Construction of supramolecular self-assembled microfibers with fluorescent properties through a modified Ionic Self-Assembly (ISA) strategy. Chem. A Eur. J. 2013, 19, 1076–1081. [Google Scholar] [CrossRef]
- Faul, C.F.J.; Antonietti, M. Ionic self-assembly: Facile synthesis of supramolecular materials. Adv. Mater. 2003, 15, 673–683. [Google Scholar] [CrossRef]
- Velichkova, R.S.; Christova, D.C. Amphiphilic polymers from macromonomers and telechelics. Prog. Polym. Sci. 1995, 20, 819–887. [Google Scholar] [CrossRef]
- Jun, H.; Le Kim, T.H.; Han, S.W.; Seo, M.; Kim, J.W.; Nam, Y.S. Polyglycerol-poly(ε-caprolactone) block copolymer as a new semi-solid polymeric emulsifier to stabilize O/W nanoemulsions. Colloid Polym. Sci. 2015, 293, 2949–2956. [Google Scholar] [CrossRef]
- Kronberg, B.; Holmberg, K.; Lindman, B. Types of surfactants, their synthesis, and applications. In Surface Chemistry of Surfactants and Polymers; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2014; pp. 1–47. [Google Scholar] [CrossRef]
- Laschewsky, A. Molecular concepts, self-organisation and properties of polysoaps. In Polysoaps/Stabilizers/Nitrogen-15 NMR; Springer: Berlin/Heidelberg, Germany, 1995; pp. 1–86. [Google Scholar] [CrossRef]
- Tadros, T. Polymeric surfactants in disperse systems. Adv. Colloid Interface Sci. 2009, 147–148, 281–299. [Google Scholar] [CrossRef] [PubMed]
- Khristov, K.; Czarnecki, J. Emulsion films stabilized by natural and polymeric surfactants. Curr. Opin. Colloid Interface Sci. 2010, 15, 324–329. [Google Scholar] [CrossRef]
- Wang, Y.; Fang, J.; Cheng, D.; Wang, Y.; Shuai, X. A pH-sensitive micelle for codelivery of siRNA and doxorubicin to hepatoma cells. Polymer 2014, 55, 3217–3226. [Google Scholar] [CrossRef]
- Yi, Y.; Lin, G.; Chen, S.; Liu, J.; Zhang, H.; Mi, P. Polyester micelles for drug delivery and cancer theranostics: Current achievements, progresses and future perspectives. Mater. Sci. Eng. C 2018, 83, 218–232. [Google Scholar] [CrossRef]
- Solov’eva, A.B.; Aksenova, N.A.; Glagolev, N.N.; Melik-Nubarov, N.S.; Ivanov, A.V.; Volkov, V.I.; Chernyak, A.V. Amphiphilic polymers in photodynamic therapy. Russ. J. Phys. Chem. B 2012, 6, 433–440. [Google Scholar] [CrossRef]
- Raffa, P.; Broekhuis, A.A.; Picchioni, F. Amphiphilic copolymers based on PEG-acrylate as surface active water viscosifiers: Towards new potential systems for enhanced oil recovery. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Raffa, P.; Broekhuis, A.A.; Picchioni, F. Polymeric surfactants for enhanced oil recovery: A review. J. Pet. Sci. Eng. 2016, 145, 723–733. [Google Scholar] [CrossRef] [Green Version]
- Barakat, Y.; Gendy, T.S.; Basily, I.K.; Mohamad, A.I. Polymeric surfactants for enhanced oil recovery. Part II—The hlb-cmc relationship of ethoxylated alkylphenol-formaldehyde polymeric surfactants. Br. Polym. J. 1989, 21, 451–457. [Google Scholar] [CrossRef]
- Migliore, N.; Picchioni, F.; Raffa, P. The effect of macromolecular structure on the rheology and surface properties of amphiphilic random polystyrene-r-poly(meth)acrylate copolymers prepared by RDRP. Soft Matter 2020, 16, 2836–2846. [Google Scholar] [CrossRef] [Green Version]
- Wever, D.A.Z.; Raffa, P.; Picchioni, F.; Broekhuis, A.A. Acrylamide homopolymers and acrylamide–N-isopropylacrylamide block copolymers by atomic transfer radical polymerization in water. Macromolecules 2012, 45, 4040–4045. [Google Scholar] [CrossRef] [Green Version]
- Raffa, P.; Brandenburg, P.; Wever, D.A.Z.; Broekhuis, A.A.; Picchioni, F. Polystyrene–poly(sodium methacrylate) amphiphilic block copolymers by ATRP: Effect of structure, pH, and ionic strength on rheology of aqueous solutions. Macromolecules 2013, 46, 7106–7111. [Google Scholar] [CrossRef] [Green Version]
- Chiefari, J.; Chong, Y.K.; Ercole, F.; Krstina, J.; Jeffery, J.; Le, T.P.T.; Mayadunne, R.T.A.; Meijs, G.F.; Moad, C.L.; Moad, G.; et al. Living free-radical polymerization by reversible addition−fragmentation chain transfer: The RAFT process. Macromolecules 1998, 31, 5559–5562. [Google Scholar] [CrossRef]
- Chong, Y.K.; Le, T.P.T.; Moad, G.; Rizzardo, E.; Thang, S.H. A more versatile route to block copolymers and other polymers of complex architecture by living radical polymerization: The RAFT process. Macromolecules 1999, 32, 2071–2074. [Google Scholar] [CrossRef]
- Nicolas, J.; Guillaneuf, Y.; Lefay, C.; Bertin, D.; Gigmes, D.; Charleux, B. Nitroxide-mediated polymerization. Prog. Polym. Sci. 2013, 38, 63–235. [Google Scholar] [CrossRef]
- Yeow, J.; Chapman, R.; Gormley, A.J.; Boyer, C. Up in the air: Oxygen tolerance in controlled/living radical polymerisation. Chem. Soc. Rev. 2018, 47, 4357–4387. [Google Scholar] [CrossRef]
- Araya-Hermosilla, E.; Catalán-Toledo, J.; Muñoz-Suescun, F.; Oyarzun-Ampuero, F.; Raffa, P.; Polgar, L.M.; Picchioni, F.; Moreno-Villoslada, I. Totally organic redox-active pH-sensitive nanoparticles stabilized by amphiphilic aromatic polyketones. J. Phys. Chem. B 2018, 122, 1747–1755. [Google Scholar] [CrossRef]
- Araya-Hermosilla, R.; Broekhuis, A.A.; Picchioni, F. Reversible polymer networks containing covalent and hydrogen bonding interactions. Eur. Polym. J. 2014, 50, 127–134. [Google Scholar] [CrossRef]
- Araya-Hermosilla, R.; Fortunato, G.; Pucci, A.; Raffa, P.; Polgar, L.; Broekhuis, A.A.; Pourhossein, P.; Lima, G.M.R.; Beljaars, M.; Picchioni, F. Thermally reversible rubber-toughened thermoset networks via Diels–Alder chemistry. Eur. Polym. J. 2016, 74, 229–240. [Google Scholar] [CrossRef]
- Araya-Hermosilla, R.; Lima, G.M.R.; Raffa, P.; Fortunato, G.; Pucci, A.; Flores, M.E.; Moreno-Villoslada, I.; Broekhuis, A.A.; Picchioni, F. Intrinsic self-healing thermoset through covalent and hydrogen bonding interactions. Eur. Polym. J. 2016, 81, 186–197. [Google Scholar] [CrossRef]
- Araya-Hermosilla, R.; Pucci, A.; Araya-Hermosilla, E.; Pescarmona, P.P.; Raffa, P.; Polgar, L.M.; Moreno-Villoslada, I.; Flores, M.; Fortunato, G.; Broekhuis, A.A.; et al. An easy synthetic way to exfoliate and stabilize MWCNTs in a thermoplastic pyrrole-containing matrix assisted by hydrogen bonds. RSC Adv. 2016, 6, 85829–85837. [Google Scholar] [CrossRef] [Green Version]
- Araya-Hermosilla, R.; Pucci, A.; Raffa, P.; Santosa, D.; Pescarmona, P.P.; Gengler, Y.N.R.; Rudolf, P.; Moreno-Villoslada, I.; Picchioni, F. Electrically-responsive reversible polyketone/MWCNT network through diels-alder chemistry. Polymers 2018, 10, 1076. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Migliore, N.; Polgar, M.L.; Araya-Hermosilla, R.; Picchioni, F.; Raffa, P.; Pucci, A. Effect of the polyketone aromatic pendent groups on the electrical conductivity of the derived MWCNTs-based nanocomposites. Polymers 2018, 10, 618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raffa, P.; Wever, D.A.Z.; Picchioni, F.; Broekhuis, A.A. Polymeric surfactants: Synthesis, properties, and links to applications. Chem. Rev. 2015, 115, 8504–8563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Broekhuis, A.A.; Picchioni, F. Aqueous polymer emulsions by chemical modifications of thermosetting alternating polyketones. J. Appl. Polym. Sci. 2007, 106, 3237–3247. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Broekhuis, A.A.; Stuart, M.C.A.; Picchioni, F. Polymeric amines by chemical modifications of alternating aliphatic polyketones. J. Appl. Polym. Sci. 2008, 107, 262–271. [Google Scholar] [CrossRef]
- Hamarneh, A.I.; Heeres, H.J.; Broekhuis, A.A.; Sjollema, K.A.; Zhang, Y.; Picchioni, F. Use of soy proteins in polyketone-based wood adhesives. Int. J. Adhes. Adhes. 2010, 30, 626–635. [Google Scholar] [CrossRef]
- Toncelli, C.; Schoonhoven, M.-J.; Broekhuis, A.A.; Picchioni, F. Paal-Knorr kinetics in waterborne polyketone-based formulations as modulating cross-linking tool in electrodeposition coatings. Mater. Des. 2016, 108, 718–724. [Google Scholar] [CrossRef]
- Drain, C.M.; Hupp, J.T.; Suslick, K.S.; Wasielewski, M.R.; Chen, X. A perspective on four new porphyrin-based functional materials and devices. J. Porphyr. Phthalocyanines 2002, 06, 243–258. [Google Scholar] [CrossRef]
- Gouterman, M. Study of the effects of substitution on the absorption spectra of porphin. J. Chem. Phys. 1959, 30, 1139–1161. [Google Scholar] [CrossRef]
- Gouterman, M. Spectra of porphyrins. J. Mol. Spectrosc. 1961, 6, 138–163. [Google Scholar] [CrossRef]
- Prigorchenko, E.; Ustrnul, L.; Borovkov, V.; Aav, R. Heterocomponent ternary supramolecular complexes of porphyrins: A review. J. Porphyr. Phthalocyanines 2019, 23, 1308–1325. [Google Scholar] [CrossRef] [Green Version]
- Schwab, A.D.; Smith, D.E.; Bond-Watts, B.; Johnston, D.E.; Hone, J.; Johnson, A.T.; de Paula, J.C.; Smith, W.F. Photoconductivity of self-assembled porphyrin nanorods. Nano Lett. 2004, 4, 1261–1265. [Google Scholar] [CrossRef]
- Komagoe, K.; Tamagake, K.; Katsu, T. The influence of aggregation of porphyrins on the efficiency of photogeneration of hydrogen peroxide in aqueous solution. Chem. Pharm. Bull. 2006, 54, 1004–1009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lapes, M.; Petera, J.; Jirsa, M. Photodynamic therapy of cutaneous metastases of breast cancer after local application of meso-tetra-(para-sulphophenyl)-porphin (TPPS4). J. Photochem. Photobiol. B 1996, 36, 205–207. [Google Scholar] [CrossRef]
- Binder, S.; Kolarova, H.; Tomankova, K.; Bajgar, R.; Daskova, A.; Mosinger, J. Phototoxic effect of TPPS4 and MgTPPS4 on DNA fragmentation of HeLa cells. Toxicol. Vitr. 2011, 25, 1169–1172. [Google Scholar] [CrossRef]
- Canaparo, R.; Varchi, G.; Ballestri, M.; Foglietta, F.; Sotgiu, G.; Guerrini, A.; Francovich, A.; Civera, P.; Frairia, R.; Serpe, L. Polymeric nanoparticles enhance the sonodynamic activity of meso-tetrakis (4-sulfonatophenyl) porphyrin in an in vitro neuroblastoma model. Int. J. Nanomed. 2013, 8, 4247–4263. [Google Scholar] [CrossRef]
- Grinstaff, M.W.; Hill, M.G.; Labinger, J.A.; Gray, H.B. Mechanism of catalytic oxygenation of alkanes by halogenated iron porphyrins. Science 1994, 264, 1311. [Google Scholar] [CrossRef]
- Tian, J.; Liu, S.; Liu, Z.; Yang, J.; Zhu, J.; Qiao, M.; Hu, X. Fluorescence quenching and spectrophotometric methods for the determination of daunorubicin with meso-tera (4-sulphophenyl) porphyrin as probe. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 120, 7–13. [Google Scholar] [CrossRef]
- Caselli, M. Porphyrin-based electrostatically self-assembled multilayers as fluorescent probes for mercury(ii) ions: A study of the adsorption kinetics of metal ions on ultrathin films for sensing applications. RSC Adv. 2015, 5, 1350–1358. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Li, M.; Liu, M.; Zhang, Y.; Wu, C.; Zhang, Y. Porphyrin-functionalized porous polysulfone membrane towards an optical sensor membrane for sorption and detection of cadmium(II). J. Hazard. Mater. 2016, 301, 233–241. [Google Scholar] [CrossRef]
- Ishida, Y.; Shimada, T.; Masui, D.; Tachibana, H.; Inoue, H.; Takagi, S. Efficient excited energy transfer reaction in clay/porphyrin complex toward an artificial light-harvesting system. J. Am. Chem. Soc. 2011, 133, 14280–14286. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, L.P.F.; Borissevitch, I.E. On the dynamics of the TPPS4 aggregation in aqueous solutions: Successive formation of H and J aggregates. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2006, 63, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Egawa, Y.; Hayashida, R.; Anzai, J.-i. pH-Induced Interconversion between J-Aggregates and H-Aggregates of 5,10,15,20-Tetrakis(4-sulfonatophenyl)porphyrin in Polyelectrolyte Multilayer Films. Langmuir 2007, 23, 13146–13150. [Google Scholar] [CrossRef] [PubMed]
- Koti, A.S.R.; Taneja, J.; Periasamy, N. Control of coherence length and aggregate size in the J-aggregate of porphyrin. Chem. Phys. Lett. 2003, 375, 171–176. [Google Scholar] [CrossRef]
- Toncelli, C.; Pino-Pinto, J.P.; Sano, N.; Picchioni, F.; Broekhuis, A.A.; Nishide, H.; Moreno-Villoslada, I. Controlling the aggregation of 5,10,15,20-tetrakis-(4-sulfonatophenyl)-porphyrin by the use of polycations derived from polyketones bearing charged aromatic groups. Dye. Pigment. 2013, 98, 51–63. [Google Scholar] [CrossRef]
- Moreno-Villoslada, I.; Murakami, T.; Nishide, H. Comment on “J- and H-aggregates of 5,10,15,20-tetrakis-(4−sulfonatophenyl)-porphyrin and interconversion in PEG-b-P4VP micelles”. Biomacromolecules 2009, 10, 3341–3342. [Google Scholar] [CrossRef]
- Kubát, P.; Lang, K.; Janda, P.; Anzenbacher, P. Interaction of porphyrins with a dendrimer template: Self-aggregation controlled by pH. Langmuir 2005, 21, 9714–9720. [Google Scholar] [CrossRef] [Green Version]
- Paulo, P.M.R.; Costa, S.M.B. Non-covalent dendrimer–porphyrin interactions: The intermediacy of H-aggregates? Photochem. Photobiol. Sci. 2003, 2, 597–604. [Google Scholar] [CrossRef]
- Zhao, L.; Ma, R.; Li, J.; Li, Y.; An, Y.; Shi, L. J- and H-aggregates of 5,10,15,20-tetrakis-(4-sulfonatophenyl)-porphyrin and interconversion in PEG-b-P4VP micelles. Biomacromolecules 2008, 9, 2601–2608. [Google Scholar] [CrossRef]
- Andrade, S.M.; Teixeira, R.; Costa, S.M.B.; Sobral, A.J.F.N. Self-aggregation of free base porphyrins in aqueous solution and in DMPC vesicles. Biophys. Chem. 2008, 133, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.J.; Hupp, J.T.; Nguyen, S.T. Growth of narrowly dispersed porphyrin nanowires and their hierarchical assembly into macroscopic columns. J. Am. Chem. Soc. 2008, 130, 9632–9633. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; An, Y.; Chen, X.; Xiong, D.a.; Li, Y.; Huang, N.; Shi, L. Chiral polymeric micelles from electrostatic assembly between achiral porphyrins and block copolymers. Macromol. Rapid Commun. 2008, 29, 214–218. [Google Scholar] [CrossRef]
- Zhao, L.; Qu, R.; Li, A.; Ma, R.; Shi, L. Cooperative self-assembly of porphyrins with polymers possessing bioactive functions. Chem. Commun. 2016, 52, 13543–13555. [Google Scholar] [CrossRef] [PubMed]
- Ruthard, C.; Maskos, M.; Kolb, U.; Gröhn, F. Polystyrene sulfonate–porphyrin assemblies: Influence of polyelectrolyte and porphyrin structure. J. Phys. Chem. B 2011, 115, 5716–5729. [Google Scholar] [CrossRef]
- Castriciano, M.A.; Romeo, A.; Angelini, N.; Micali, N.; Longo, A.; Mazzaglia, A.; Scolaro, L.M. Structural features of meso-tetrakis(4-carboxyphenyl)porphyrin interacting with amino-terminated poly(propylene oxide). Macromolecules 2006, 39, 5489–5496. [Google Scholar] [CrossRef]
- Shen, L.; Zhao, L.; Qu, R.; Huang, F.; Gao, H.; An, Y.; Shi, L. Complex micelles with the bioactive function of reversible oxygen transfer. Nano Res. 2015, 8, 491–501. [Google Scholar] [CrossRef]
- Wang, M.; Yan, F.; Zhao, L.; Zhang, Y.; Sorci, M. Preparation and characterization of a pH-responsive membrane carrier for meso-tetraphenylsulfonato porphyrin. RSC Adv. 2017, 7, 1687–1696. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Zhao, L.; Li, S.; Ye, H.; An, H.; Zhang, Y. pH-responsive ethylene vinyl alcohol copolymer membrane based on porphyrin supramolecular self-assembly. RSC Adv. 2016, 6, 10704–10712. [Google Scholar] [CrossRef]
- Fagadar-Cosma, E.; Vlascici, D.; Birdeanu, M.; Fagadar-Cosma, G. Novel fluorescent pH sensor based on 5-(4-carboxy-phenyl)-10,15,20-tris(phenyl)-porphyrin. Arab. J. Chem. 2019, 12, 1587–1594. [Google Scholar] [CrossRef] [Green Version]
- Thyagarajan, S.; Leiding, T.; Årsköld, S.P.; Cheprakov, A.V.; Vinogradov, S.A. Highly non-planar dendritic porphyrin for pH sensing: Observation of porphyrin monocation. Inorg. Chem. 2010, 49, 9909–9920. [Google Scholar] [CrossRef] [Green Version]
- Igarashi, S.; Kuwae, K.; Yotsuyanagi, T. Optical pH sensor of electrostatically immobilized porphyrin on the surface of sulfonated-polystyrene. Anal. Sci. 1994, 10, 821–822. [Google Scholar] [CrossRef] [Green Version]
- Araya-Hermosilla, E.; Roscam Abbing, M.; Catalán-Toledo, J.; Oyarzun-Ampuero, F.; Pucci, A.; Raffa, P.; Picchioni, F.; Moreno-Villoslada, I. Synthesis of tuneable amphiphilic-modified polyketone polymers, their complexes with 5,10,15,20-tetrakis-(4-sulfonatophenyl)porphyrin, and their role in the photooxidation of 1,3,5-triphenylformazan confined in polymeric nanoparticles. Polymer 2019, 167, 215–223. [Google Scholar] [CrossRef]
- Mul, W.P.; Dirkzwager, H.; Broekhuis, A.A.; Heeres, H.J.; van der Linden, A.J.; Guy Orpen, A. Highly active, recyclable catalyst for the manufacture of viscous, low molecular weight, CO–ethene–propene-based polyketone, base component for a new class of resins. Inorg. Chim. Acta 2002, 327, 147–159. [Google Scholar] [CrossRef] [Green Version]
- Aydinoglu, S.; Biver, T.; Ceccarini, A.; Secco, F.; Venturini, M. Gold(III) extraction and recovery and gold(III)/copper(II) separation using micelles. Colloids Surf. A Physicochem. Eng. Asp. 2015, 482, 324–328. [Google Scholar] [CrossRef] [Green Version]
- Dai, S.; Ravi, P.; Tam, K.C. pH-Responsive polymers: Synthesis, properties and applications. Soft Matter 2008, 4, 435–449. [Google Scholar] [CrossRef]
- Jeong, B.; Gutowska, A. Lessons from nature: Stimuli-responsive polymers and their biomedical applications. Trends Biotechnol. 2002, 20, 305–311. [Google Scholar] [CrossRef]
- Ghorbani Gorji, S.; Ghorbani Gorji, E.; Mohammadifar, M.A. Effect of pH on turbidity, size, viscosity and the shape of sodium caseinate aggregates with light scattering and rheometry. J. Food Sci. Technol. 2015, 52, 1820–1824. [Google Scholar] [CrossRef]
- Wang, X.; Chai, Z.; Ma, R.; Zhao, L.; Zhang, Z.; An, Y.; Shi, L. Enhancement of the photostability and photoactivity of metallo-meso-5,10,15,20-tetrakis-(4-sulfonatophenyl)porphyrins by polymeric micelles. J. Colloid Interface Sci. 2012, 388, 80–85. [Google Scholar] [CrossRef]
- Li, A.; Zhao, L.; Hao, J.; Ma, R.; An, Y.; Shi, L. Aggregation behavior of the template-removed 5,10,15,20-tetrakis(4-sulfonatophenyl)porphyrin chiral array directed by poly(ethylene glycol)-block-poly(l-lysine). Langmuir 2014, 30, 4797–4805. [Google Scholar] [CrossRef]
- Maiti, N.C.; Mazumdar, S.; Periasamy, N. J- and H-aggregates of porphyrin−surfactant complexes: Time-resolved fluorescence and other spectroscopic studies. J. Phys. Chem. B 1998, 102, 1528–1538. [Google Scholar] [CrossRef]
- Xu, W.; Guo, H.; Akins, D.L. Aggregation of tetrakis(p-sulfonatophenyl)porphyrin within modified mesoporous MCM-41. J. Phys. Chem. B 2001, 105, 1543–1546. [Google Scholar] [CrossRef]
- Ma, H.-L.; Jin, W.-J. Studies on the effects of metal ions and counter anions on the aggregate behaviors of meso-tetrakis(p-sulfonatophenyl)porphyrin by absorption and fluorescence spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2008, 71, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Tardajos, M.; Pino-Pinto, J.P.; Díaz-Soto, C.; Flores, M.E.; Gallardo, A.; Elvira, C.; Reinecke, H.; Nishide, H.; Moreno-Villoslada, I. Confinement of 5,10,15,20-tetrakis-(4-sulfonatophenyl)-porphyrin in novel poly(vinylpyrrolidone)s modified with aromatic amines. Dye. Pigment. 2013, 99, 759–770. [Google Scholar] [CrossRef] [Green Version]
- Feng, X.; Chen, L.; Dong, Y.; Jiang, D. Porphyrin-based two-dimensional covalent organic frameworks: Synchronized synthetic control of macroscopic structures and pore parameters. Chem. Commun. 2011, 47, 1979–1981. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I.; Mukherjee, K.; Shoukat, R.; Dong, H. A review on pH sensitive materials for sensors and detection methods. Microsyst. Technol. 2017, 23, 4391–4404. [Google Scholar] [CrossRef]












© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Araya-Hermosilla, E.; Moreno-Villoslada, I.; Araya-Hermosilla, R.; Flores, M.E.; Raffa, P.; Biver, T.; Pucci, A.; Picchioni, F.; Mattoli, V. pH-Responsive Polyketone/5,10,15,20-Tetrakis-(Sulfonatophenyl)Porphyrin Supramolecular Submicron Colloidal Structures. Polymers 2020, 12, 2017. https://0-doi-org.brum.beds.ac.uk/10.3390/polym12092017
Araya-Hermosilla E, Moreno-Villoslada I, Araya-Hermosilla R, Flores ME, Raffa P, Biver T, Pucci A, Picchioni F, Mattoli V. pH-Responsive Polyketone/5,10,15,20-Tetrakis-(Sulfonatophenyl)Porphyrin Supramolecular Submicron Colloidal Structures. Polymers. 2020; 12(9):2017. https://0-doi-org.brum.beds.ac.uk/10.3390/polym12092017
Chicago/Turabian StyleAraya-Hermosilla, Esteban, Ignacio Moreno-Villoslada, Rodrigo Araya-Hermosilla, Mario E. Flores, Patrizio Raffa, Tarita Biver, Andrea Pucci, Francesco Picchioni, and Virgilio Mattoli. 2020. "pH-Responsive Polyketone/5,10,15,20-Tetrakis-(Sulfonatophenyl)Porphyrin Supramolecular Submicron Colloidal Structures" Polymers 12, no. 9: 2017. https://0-doi-org.brum.beds.ac.uk/10.3390/polym12092017