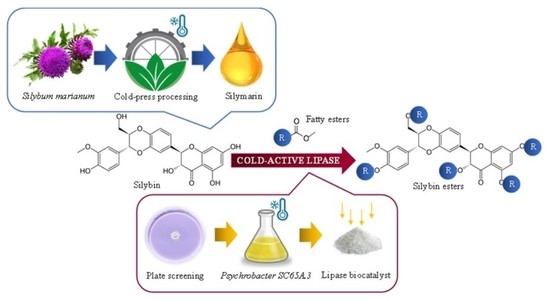
Cold-Active Lipase-Based Biocatalysts for Silymarin Valorization through Biocatalytic Acylation of Silybin
Abstract
:1. Introduction
2. Results and Discussion
2.1. Evaluation of Psychrobacter SC65A.3 Lipolytic Activity on Different Substrates
2.2. Characterization of the Immobilized Cold-Active Lipase
2.3. Cold-Active Lipase Catalyzing Silybin Acylation
3. Materials and Methods
3.1. Chemicals and Solutions
3.2. Plate Screening Assays
3.3. Preparation of the Protein Extract
3.4. Enzyme Assay
3.5. Lipase Immobilization
3.5.1. Preparation of the Immobilized Lipase Biocatalyst
3.5.2. Characterization of the Immobilized Lipase Biocatalyst
- mi—initial protein amount (after concentration of the extracellular extract);
- ms—protein amount in the supernatant after immobilization step;
- LAimmob—lipase activity of the immobilized protein fraction;
- LAi—lipase activity of the solution used for immobilization;
- LAs—lipase activity of the supernatant after immobilization.
3.6. Biocatalytic System for Silybin Acylation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Flora, K.; Hahn, M.; Rosen, H.; Benner, K. Milk Thistle (Silybum marianum) for the Therapy of Liver Disease. Am. J. Gastroenterol. 1998, 93, 139–143. [Google Scholar] [CrossRef]
- Kuki, Á.; Nagy, L.; Deák, G.; Nagy, M.; Zsuga, M.; Kéki, S. Identification of Silymarin Constituents: An Improved HPLC–MS Method. Chromatographia 2011, 75, 175–180. [Google Scholar] [CrossRef]
- Begum, S.A.; Sahai, M.; Ray, A.B. Non-conventional Lignans: Coumarinolignans, Flavonolignans, and Stilbenolignans. Fortschr. Chem. Org. Nat. 2010, 93, 1–70. [Google Scholar]
- Csupor, D.; Csorba, A.; Hohmann, J. Recent advances in the analysis of flavonolignans of Silybum marianum. J. Pharm. Biomed. Anal. 2016, 130, 301–317. [Google Scholar] [CrossRef]
- Vostálová, J.; Tinková, E.; Biedermann, D.; Kosina, P.; Ulrichová, J.; Svobodová, A.R. Skin Protective Activity of Silymarin and its Flavonolignans. Molecules 2019, 24, 1022. [Google Scholar] [CrossRef] [Green Version]
- Bijak, M. Silybin, a Major Bioactive Component of Milk Thistle (Silybum marianum L. Gaernt.)—Chemistry, Bioavailability, and Metabolism. Molecules 2017, 22, 1942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biedermann, D.; Vavříková, E.; Cvak, L.; Křen, V. Chemistry of silybin. Nat. Prod. Rep. 2014, 31, 1138–1157. [Google Scholar] [CrossRef]
- Drouet, S.; Doussot, J.; Garros, L.; Mathiron, D.; Bassard, S.; Favre-Réguillon, A.; Molinié, R.; Lainé, É.; Hano, C. Selective Synthesis of 3-O-Palmitoyl-Silybin, a New-to-Nature Flavonolignan with Increased Protective Action against Oxidative Damages in Lipophilic Media. Molecules 2018, 23, 2594. [Google Scholar] [CrossRef] [Green Version]
- Duan, L.; Carrier, D.J.; Clausen, E.C. Silymarin Extraction from Milk Thistle Using Hot Water. Appl. Biochem. Biotechnol. 2004, 114, 559–568. [Google Scholar] [CrossRef]
- Theodosiou, E.; Purchartová, K.; Stamatis, H.; Kolisis, F.; Křen, V. Bioavailability of silymarin flavonolignans: Drug formulations and biotransformation. Phytochem. Rev. 2013, 13, 1–18. [Google Scholar] [CrossRef]
- Duran, D.; Ötleş, S.; Karasulu, E. Determination amount of silymarin and pharmaceutical products from milk thistle waste obtained from cold press. Acta Pharm. Sci. 2019, 57, 85. [Google Scholar] [CrossRef]
- Fidrus, E.; Ujhelyi, Z.; Fehér, P.; Hegedűs, C.; Janka, E.A.; Paragh, G.; Vasas, G.; Bácskay, I.; Remenyik, É. Silymarin: Friend or Foe of UV Exposed Keratinocytes? Molecules 2019, 24, 1652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hackett, E.S.; Twedt, D.C.; Gustafson, D.L. Milk Thistle and Its Derivative Compounds: A Review of Opportunities for Treatment of Liver Disease. J. Vet. Intern. Med. 2013, 27, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Di Costanzo, A.; Angelico, R. Formulation Strategies for Enhancing the Bioavailability of Silymarin: The State of the Art. Molecules 2019, 24, 2155. [Google Scholar] [CrossRef] [Green Version]
- Di Fabio, G.; Zarrelli, A.; Romanucci, V.; Della Greca, M.; De Napoli, L.; Previtera, L. New Silybin Scaffold for Chemical Diversification: Synthesis of Novel 23-Phosphodiester Silybin Conjugates. Synlett 2012, 24, 45–48. [Google Scholar] [CrossRef] [Green Version]
- Chambers, C.S.; Biedermann, D.; Valentová, K.; Petrásková, L.; Viktorová, J.; Kuzma, M.; Křen, V. Preparation of Retinoyl-Flavonolignan Hybrids and Their Antioxidant Properties. Antioxidants 2019, 8, 236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kesharwani, S.S.; Jain, V.; Dey, S.; Sharma, S.; Mallya, P.; Kumar, V.A. An Overview of Advanced Formulation and Nanotechnology-based Approaches for Solubility and Bioavailability Enhancement of Silymarin. J. Drug Deliv. Sci. Technol. 2020, 60, 102021. [Google Scholar] [CrossRef]
- Xanthakis, E.; Theodosiou, E.; Magkouta, S.; Stamatis, H.; Loutrari, H.; Roussos, C.; Kolisis, F. Enzymatic transformation of flavonoids and terpenoids: Structural and functional diversity of the novel derivatives. Pure Appl. Chem. 2010, 82, 1–16. [Google Scholar] [CrossRef]
- Vavříková, E.; Gavezzotti, P.; Purchartová, K.; Fuksová, K.; Biedermann, D.; Kuzma, M.; Riva, S.; Křen, V. Regioselective Alcoholysis of Silychristin Acetates Catalyzed by Lipases. Int. J. Mol. Sci. 2015, 16, 11983–11995. [Google Scholar] [CrossRef] [Green Version]
- Brenchley, J. Psychrophilic microorganisms and their cold-active enzymes. J. Ind. Microbiol. Biotechnol. 1996, 17, 432–437. [Google Scholar] [CrossRef]
- Mangiagalli, M.; Brocca, S.; Orlando, M.; Lotti, M. The “cold revolution”. Present and future applications of cold-active enzymes and ice-binding proteins. New Biotechnol. 2020, 55, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Santiago, M.; Ramírez, C.; Zamora, R.; Parra, L.P. Discovery, Molecular Mechanisms, and Industrial Applications of Cold-Active Enzymes. Front. Microbiol. 2016, 7, 1408. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, M. Cold active lipases—An update. Front. Life Sci. 2016, 9, 226–238. [Google Scholar] [CrossRef] [Green Version]
- Persoiu, A.; Lauritzen, S.E. Scarisoara Ice Cave. In Ice Caves, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 25.3.2.3; pp. 520–527. [Google Scholar]
- Paun, V.I.; Lavin, P.; Chifiriuc, M.C.; Purcarea, C. First report on antibiotic resistance and antimicrobial activity of bacterial isolates from 13,000-year old cave ice core. Sci. Rep. 2021, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Paun, V.I.; Icaza, G.; Lavin, P.; Marin, C.; Tudorache, A.; Perşoiu, A.; Dorador, C.; Purcarea, C. Total and Potentially Active Bacterial Communities Entrapped in a Late Glacial Through Holocene Ice Core From Scarisoara Ice Cave, Romania. Front. Microbiol. 2019, 10, 1193. [Google Scholar] [CrossRef]
- de María, P.D.; Carboni-Oerlemans, C.; Tuin, B.; Bargeman, G.; van der Meer, A.; van Gemert, R. Biotechnological applications of Candida antarctica lipase A: State-of-the-art. J. Mol. Catal. B Enzym. 2005, 37, 36–46. [Google Scholar] [CrossRef]
- Stergiou, P.-Y.; Foukis, A.; Filippou, M.; Koukouritaki, M.; Parapouli, M.; Theodorou, L.G.; Hatziloukas, E.; Afendra, A.; Pandey, A.; Papamichael, E.M. Advances in lipase-catalyzed esterification reactions. Biotechnol. Adv. 2013, 31, 1846–1859. [Google Scholar] [CrossRef]
- Das, U.N. Essential fatty acids: Biochemistry, physiology and pathology. Biotechnol. J. 2006, 1, 420–439. [Google Scholar] [CrossRef]
- Lawerence, G.D. The Fats of Life. Essential Fatty Acids in Health and Disease; Rutgers University Press: New Brunswick, NJ, USA, 2010; Chapter 2; pp. 15–20. [Google Scholar]
- Gandhi, N.N.; Patil, N.S.; Sawant, S.B.; Joshi, J.B.; Wangikar, P.P.; Mukesh, D. Lipase-Catalyzed Esterification. Catal. Rev. 2000, 42, 439–480. [Google Scholar] [CrossRef]
- Illanes, A. Chimioselective Esterification of Wood Sterols with Lipases. In Enzyme Biocatalysis; Springer: New York, NY, USA, 2008; Chapter 6.3; pp. 292–308. [Google Scholar]
- Ramnath, L.; Sithole, B.; Govinden, R. Identification of lipolytic enzymes isolated from bacteria indigenous to Eucalyptus wood species for application in the pulping industry. Biotechnol. Rep. 2017, 15, 114–124. [Google Scholar] [CrossRef]
- Peña, S.A.; Rios, N.S.; Carballares, D.; Sánchez, C.M.; Lokha, Y.; Gonçalves, L.R.B.; Fernandez-Lafuente, R. Effects of Enzyme Loading and Immobilization Conditions on the Catalytic Features of Lipase From Pseudomonas fluorescens Immobilized on Octyl-Agarose Beads. Front. Bioeng. Biotechnol. 2020, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta (BBA) Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef] [Green Version]
- Stoia, M.; Istratie, R.; Păcurariu, C. Investigation of magnetite nanoparticles stability in air by thermal analysis and FTIR spectroscopy. J. Therm. Anal. Calorim. 2016, 125, 1185–1198. [Google Scholar] [CrossRef]
- Liu, X.; Guan, Y.; Shen, R.; Liu, H. Immobilization of lipase onto micron-size magnetic beads. J. Chromatogr. B 2005, 822, 91–97. [Google Scholar] [CrossRef]
- Xu, Y.-Q.; Zhou, G.-W.; Wu, C.-C.; Li, T.-D.; Song, H.-B. Improving adsorption and activation of the lipase immobilized in amino-functionalized ordered mesoporous SBA-15. Solid State Sci. 2011, 13, 867–874. [Google Scholar] [CrossRef]
- Rehm, S.; Trodler, P.; Pleiss, J. Solvent-induced lid opening in lipases: A molecular dynamics study. Protein Sci. 2010, 19, 2122–2130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adlercreutz, P. Immobilisation and application of lipases in organic media. Chem. Soc. Rev. 2013, 42, 6406–6436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bassi, J.J.; Todero, L.M.; Lage, F.A.; Khedy, G.I.; Ducas, J.D.; Custódio, A.P.; Pinto, M.A.; Mendes, A.A. Interfacial activation of lipases on hydrophobic support and application in the synthesis of a lubricant ester. Int. J. Biol. Macromol. 2016, 92, 900–909. [Google Scholar] [CrossRef] [PubMed]
- Peña, S.A.; Rios, N.S.; Carballares, D.; Gonçalves, L.R.; Fernandez-Lafuente, R. Immobilization of lipases via interfacial activation on hydrophobic supports: Production of biocatalysts libraries by altering the immobilization conditions. Catal. Today 2021, 362, 130–140. [Google Scholar] [CrossRef]
- Neagu, S.; Preda, S.; Anastasescu, C.; Zaharescu, M.; Enache, M.; Cojoc, R. The functionalization of silica and titanate nanostructures with halotolerant proteases. Rev. Roum. Chim. 2014, 59, 97–103. [Google Scholar]
- Mach, H.; Middaugh, C.; Lewis, R.V. Statistical determination of the average values of the extinction coefficients of tryptophan and tyrosine in native proteins. Anal. Biochem. 1992, 200, 74–80. [Google Scholar] [CrossRef]
- Moreno, M.D.L.; Garcia, M.T.; Ventosa, A.; Mellado, E. Characterization of Salicola sp.  IC10, a lipase- and protease-producing extreme halophile. FEMS Microbiol. Ecol. 2009, 68, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Available online: http://www.chemicell.com/home/index.html (accessed on 14 November 2021).
- Lite, C.; Ion, S.; Tudorache, M.; Zgura, I.; Galca, A.C.; Enache, M.; Maria, G.-M.; Parvulescu, V.I. Alternative lignopolymer-based composites useful as enhanced functionalized support for enzymes immobilization. Catal. Today 2020, 379, 222–229. [Google Scholar] [CrossRef]
- Hill, A.; Karboune, S.; Mateo, C. Investigating and optimizing the immobilization of levansucrase for increased transfructosylation activity and thermal stability. Process. Biochem. 2017, 61, 63–72. [Google Scholar] [CrossRef]






| Recovery of Lipase Activity (%) | Immobilization Yield (%) | Protein Loading (%) | |
|---|---|---|---|
| S1 | 82 ± 5.1 | 80 ± 4.5 | 99 ± 5.3 |
| S2 | 128 ± 6.3 | 84 ± 4.7 | 99 ± 4.8 |
| S3 | 80 ± 5.4 | 76 ± 4.3 | 93 ± 4.6 |
| S4 | 82 ± 5.5 | 74 ± 4.8 | 96 + 4.9 |
| S5 | 105 ± 5.1 | 89 ± 4.0 | 90 ± 4.3 |
| Biocatalyst | Km (mM) | kcat (min−1) | kcat/Km (mM−1 min−1) | |||
|---|---|---|---|---|---|---|
| 25 °C | 37 °C | 25 °C | 37 °C | 25 °C | 37 °C | |
| Free | 2.18 ± 0.15 | 1.85 ± 0.09 | 1.63 ± 0.08 | 1.95 ± 0.09 | 0.75 ± 0.05 | 1.05 ± 0.09 |
| S1 | 1.61 ± 0.08 | 1.17 ± 0.09 | 0.99 ± 0.05 | 0.97 ± 0.06 | 0.62 ± 0.04 | 0.83 ± 0.04 |
| S2 | 0.56 ± 0.04 | 0.65 ± 0.03 | 0.93 ± 0.05 | 0.99 ± 0.06 | 1.67 ± 0.09 | 1.54 ± 0.07 |
| S3 | 4.18 ± 0.21 | 1.41 ± 0.07 | 2.29 ± 0.09 | 1.25 ± 0.07 | 0.55 ± 0.03 | 0.88 ± 0.04 |
| S4 | 0.63 ± 0.03 | 0.78 ± 0.05 | 0.65 ± 0.03 | 0.92 ± 0.05 | 1.02 ± 0.09 | 1.17 ± 0.09 |
| S5 | 0.94 ± 0.05 | 0.64 ± 0.03 | 0.85 ± 0.08 | 1.2 ± 0.09 | 0.90 ± 0.05 | 1.88 ± 0.09 |
| Support | Immobilization Method | |
|---|---|---|
| S1 | sMP-COOH | EDC |
| S2 | fMP-COOH | EDC |
| S3 | sMP-NH2 | EDC |
| S4 | sMP-NH-NH2 | NaIO4 |
| S5 | MTC-NH2 | GA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gheorghita, G.R.; Paun, V.I.; Neagu, S.; Maria, G.-M.; Enache, M.; Purcarea, C.; Parvulescu, V.I.; Tudorache, M. Cold-Active Lipase-Based Biocatalysts for Silymarin Valorization through Biocatalytic Acylation of Silybin. Catalysts 2021, 11, 1390. https://0-doi-org.brum.beds.ac.uk/10.3390/catal11111390
Gheorghita GR, Paun VI, Neagu S, Maria G-M, Enache M, Purcarea C, Parvulescu VI, Tudorache M. Cold-Active Lipase-Based Biocatalysts for Silymarin Valorization through Biocatalytic Acylation of Silybin. Catalysts. 2021; 11(11):1390. https://0-doi-org.brum.beds.ac.uk/10.3390/catal11111390
Chicago/Turabian StyleGheorghita, Giulia Roxana, Victoria Ioana Paun, Simona Neagu, Gabriel-Mihai Maria, Madalin Enache, Cristina Purcarea, Vasile I. Parvulescu, and Madalina Tudorache. 2021. "Cold-Active Lipase-Based Biocatalysts for Silymarin Valorization through Biocatalytic Acylation of Silybin" Catalysts 11, no. 11: 1390. https://0-doi-org.brum.beds.ac.uk/10.3390/catal11111390